
Ultrasonographic optic nerve sheath diameter monitoring of elevated intracranial pressure: two case reports
Introduction
Increased intracranial pressure (ICP) is an acute medical event which is associated with poor clinical outcomes in various neurological diseases including, for example, cerebral venous sinus stenosis and subarachnoid hemorrhage. Direct assessment methods, such as lumbar puncture (LP) and intraventricular catheterization, are commonly used to detect ICP. However, these invasive techniques can result in complications, such as hemorrhaging and bacterial colonization (1,2), and involve pain and discomfort, especially if repeated evaluations are needed. In addition, invasive ICP monitoring is sometimes unavailable in intensive care units or to patients with certain serious conditions. Given this, non-invasive, repeatable, simple methods for assessing ICP are urgently needed. Recently, measuring the optic nerve sheath diameter (ONSD) via ultrasound has been proposed as a non-invasive clinical technique for detecting elevated ICP (3-6). However, this technique was used to evaluate the elevated ICP caused by severe traumatic brain injury and few studies reported the cases that prompt this technique could dynamically assess the efficacy of management of elevated ICP. We observed the patient with cerebral venous sinus stenosis and venous sinus thrombosis with elevated ICP whom we assessed for ONSD and ICP before and after treatment to investigate the potential of the ultrasonic ONSD technique to dynamically assess ICP changes and evaluate treatment efficacy. We present the following case in accordance with the CARE Guideline (7).
Case presentation
Case 1
The first case presented here was a 50-year-old woman who was admitted to our hospital due to amaurosis fugax and occasional mild headaches, free of dizziness or rotation, the symptoms of which had ceased for 2 years. She was seen in the department of ophthalmology, where an ophthalmoscopic exam revealed bilateral papilledema. The patient was then referred to the Neurology Clinic, where a physical examination revealed clear consciousness with flexible activity of the limbs. She underwent head computed tomography (CT) and magnetic resonance venography (MRV) of the head. The CT showed no obvious abnormalities and left internal jugular vein, sigmoid sinus, and transverse sinus were not clearly observed on MRV (Figure 1A,B). Thereafter, ultrasonic ONSD measurements of both eyes were conducted before a LP was performed. Ultrasound ONSD examinations were carried out using a Philips iU22 (Andover, MA, USA) ultrasound system with a 9-3 MHz linear array transducer. The first measurement was performed in the sagittal plane with the probe in a vertical orientation. The second measurement was performed in the transverse plane with the probe in a horizontal orientation. The ONSD was assessed bilaterally, 3 mm posterior to the orbit (Figure 1C). The final ONSD value for an individual was calculated as the average of bilateral measurements to minimize inter-ocular variability. According to our previous study, quantitative assessment of ICP was calculated using ONSD measurements by the mathematical model (ICP =−111.92+77.36 × ONSD) (5). The a predicted ICP by ONSD was 346 mmH2O. An LP was performed, which detected an opening pressure of 355 mmH2O.
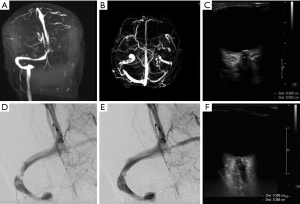
To determine the condition of the intracranial venous sinus, the patient was scheduled to undergo a digital subtraction angiography (DSA) examination. Furthermore, DSA revealed venous sinus stenosis at the junction between the right transverse sinus and the sigmoid sinus (Figure 1D). Therefore, the diagnoses of intracranial hypertension and venous sinus stenosis were established and treatment via venous sinus stenting was pursued. Postoperatively, the patient has no headache, vomiting and other adverse reactions. Further evaluation by DSA revealed obvious improvements to venous sinus stenosis (Figure 1E). Two days later, symptoms resolved and the enlarged ONSDs retracted (Figure 1F). According to the mathematical model (ICP =−111.92+77.36 × ONSD) (5), the predicted ICP was 185 mmH2O. Follow-up with LP was 180 mmH2O.
Case 2
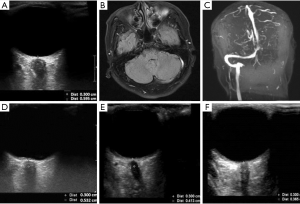
The second case presented here was of a 48-year-old man who was admitted to the Department of Neurology due to headache that had lasted for 12 h. A physical examination showed clear consciousness and flexible limbs. The patient then underwent LP and ultrasonographic assessment of ONSD. The LP revealed an opening pressure higher than 400 mmH2O, and ultrasonography showed a dilated ONSD of 5.95 mm (Figure 2A), denoting elevated ICP. The following day, the patient underwent subsequent head MRI and MRV. The MRI showed increased signal in the left sigmoid sinus and transverse sinus (Figure 2B), and MRV indicated no clear display in the left internal jugular vein, sigmoid sinus, and transverse sinus (Figure 2C). Therefore, he was definitively diagnosed with venous sinus thrombosis and treated with mannitol, to lower ICP, and anticoagulation therapies. Thereafter, the patient underwent LP at 1, 2, and 3 months after treatment, and ultrasonic ONSD measurements were also performed before the LPs to observe dynamic changes of ICP. The ONSD examination revealed a dilated ONSD of 5.32 mm (Figure 2D) and the LP detected an opening pressure of 355 mmH2O at the 1st month after treatment. At the 2nd and 3rd months of follow-up, the ONSD was 4.13 and 3.65 mm, respectively (Figure 2E,F), and the LP pressure was 210 and 180 mmH2O, respectively.
Discussion
The optic nerve sheath is the extension of the dura mater, with a transomal subarachnoid space contained cerebrospinal fluid, Hansen et al. have reported that the intra-orbital subarachnoid space, which surrounds the optic nerve, is subject to the same pressure changes as the intracranial subarachnoid space in 1996 (6). Galetta et al. found that the ONSD could be detected by B-mode ultrasonography in 1989 and confirmed with increased ICP in patients the ONSD would dilate (8). Subsequently, more and more studies confirmed a direct correlation between ONSD and invasive ICP monitoring results, with a correlation coefficient of 0.59 to 0.91 (9-13).
Ultrasonographic ONSD measurement has recently become a popular non-invasive approach for the detection of elevated ICP (13-16). Robba et al. reported that ONSD examinations more accurately approximate ICP than do transcranial Doppler ultrasonography (TCD) and other non-invasive methods studied (17). Furthermore, dynamic changes in ONSD are also strongly related to changes in ICP (18-20). Moreover, measuring ONSD via ultrasound is a repeatable and quick method, as the orbital window is easily available and uncomplicated in most patients (11,21,22). Besides, Toscano et al. illustrated the ONSD ultrasound evaluation were widely available in intensive care unit in monitoring patients with neurologic critical disease due to the effectiveness and simplicity, and demonstrating the malignant intracranial hypertension in brain death patients can be detected early (23). ONSD assessment is especially indicated when LP results are equivocal or when invasive ICP monitoring is not immediately available or is even contraindicated (19). These case reports also provide strong evidence for ultrasonographic ONSD as a reliable and meaningful method for patients who requiring repeated evaluation of ICP.
This first case presented here describes a patient with a diagnosis of venous sinus stenosis in whom the disease was progressing slowly. Her primary symptoms were visual disruptions and she did not report severe headaches. It is worth noting, based on these findings, that this type of disease may first impact eye function. In addition, the second case presented here describes a patient with venous sinus thrombosis with elevated ICP. We followed up two patients throughout the course of treatment and determined the patient’s ONSD using B-mode sonography. The two cases suggested that this noninvasive technique might allow for dynamic assessment of ICP changes throughout the patient’s clinical course and thus evaluation of the efficacy of ICP treatment. Upon admission, transorbital sonography in the two patients all revealed a markedly expanded ONSD. This finding was consistent with those of previous reports (9-12). Based on mathematical estimations (5), we determined predicted ICP that were similar to the opening pressure detected via LP. The case reports further showed that noninvasive, simple sonography could accurately and repeated estimate ICP and it would help the patient to alleviate the pain considerably compared with LP. Furthermore, after treatment, this expanded ONSD was found to return to normal, as investigated in our previous study (24).
In addition to the cases presented here, others have demonstrated the clinical efficacy of assessments of ONSD change. In an in vitro study, Hansen et al. demonstrated that ONSD changes through the controlled application of incremental, decreasing pressure steps in the subarachnoid space (25). However, few clinical studies have estimated ONSD variations following the application of treatments for elevated ICP. Launey et al. found a significant correlation between ICP and ONSD measurements before and after mannitol infusion (26). Suggesting a potential mechanism, additional studies have shown that the optic nerve sheath is quite elastic and responsive to external pressure changes (27-30). For instance, tracheal stimulation has been reported to result in simultaneous increases in both ICP and ONSD, with commensurate returns to baseline in both measures after the procedure (29). Furthermore, Hassen et al. (18) reported ONSD measurements in real time and further demonstrated dynamic changes during LP in a patient with idiopathic intracranial hypertension (IIH). And in a recent study of 84 subjects, Chen et al. reported ONSD revealed an immediate reduction from 4.13 to 4.02 mm with the decrease of cerebrospinal fluid pressure that was performed 5min pro and post LP (31). The reports strongly support that ONSD examination is a useful technique for the dynamic assessment of ICP changes.
Conclusions
These cases showed that dilated ONSDs in patients with an increased ICP resolve concomitantly with ICP after treatment. Ultrasonography of the ONSD may serve as a rapid, non-invasive, bedside technique for the dynamic and quantitative evaluation of ICP changes, especially if repeated evaluations are needed.
Acknowledgments
Funding: This work was supported by the National Natural Science Foundation of China (No. 81801707).
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Written informed consent was obtained from the patients for publication of this manuscript and any accompanying images.
References
- Amini A, Kariman H, Arhami Dolatabadi A, et al. Use of the sonographic diameter of optic nerve sheath to estimate intracranial pressure. Am J Emerg Med 2013;31:236-9. [Crossref] [PubMed]
- Dubourg J, Messerer M, Karakitsos D, et al. Individual patient data systematic review and meta-analysis of optic nerve sheath diameter ultrasonography for detecting raised intracranial pressure: protocol of the ONSD research group. Syst Rev 2013;2:62. [Crossref] [PubMed]
- Robba C, Cardim D, Tajsic T, et al. Ultrasound non-invasive measurement of intracranial pressure in neurointensive care: A prospective observational study. PLoS Med 2017;14:e1002356. [Crossref] [PubMed]
- Wang L, Feng L, Yao Y, et al. Optimal optic nerve sheath diameter threshold for the identification of elevated opening pressure on lumbar puncture in a Chinese population. PLoS One 2015;10:e0117939. [Crossref] [PubMed]
- Wang LJ, Yao Y, Feng LS, et al. Noninvasive and quantitative intracranial pressure estimation using ultrasonographic measurement of optic nerve sheath diameter. Sci Rep 2017;7:42063. [Crossref] [PubMed]
- Hansen HC, Helmke K. The subarachnoid space surrounding the optic nerves. An ultrasound study of the optic nerve sheath. Surg Radiol Anat 1996;18:323-8. [Crossref] [PubMed]
- Riley DS, Barber MS, Kienle GS, et al. CARE guidelines for case reports: explanation and elaboration document. J Clin Epidemiol 2017;89:218-35. [Crossref] [PubMed]
- Galetta S, Byrne SF, Smith JL. Echographic correlation of optic nerve sheath size and cerebrospinal fluid pressure. J Clin Neuroophthalmol 1989;9:79-82. [PubMed]
- Watanabe A, Kinouchi H, Horikoshi T, et al. Effect of intracranial pressure on the diameter of the optic nerve sheath. J Neurosurg 2008;109:255-8. [Crossref] [PubMed]
- Rajajee V, Vanaman M, Fletcher JJ, et al. Optic nerve ultrasound for the detection of raised intracranial pressure. Neurocrit Care 2011;15:506-15. [Crossref] [PubMed]
- Raffiz M, Abdullah JM. Optic nerve sheath diameter measurement: a means of detecting raised ICP in adult traumatic and non-traumatic neurosurgical patients. Am J Emerg Med 2017;35:150-3. [Crossref] [PubMed]
- Kimberly HH, Shah S, Marill K, et al. Correlation of optic nerve sheath diameter with direct measurement of intracranial pressure. Acad Emerg Med 2008;15:201-4. [Crossref] [PubMed]
- Major R, Girling S, Boyle A. Ultrasound measurement of optic nerve sheath diameter in patients with a clinical suspicion of raised intracranial pressure. Emerg Med J 2011;28:679-81. [Crossref] [PubMed]
- Soldatos T, Chatzimichail K, Papathanasiou M, et al. Optic nerve sonography: a new window for the non-invasive evaluation of intracranial pressure in brain injury. Emerg Med J 2009;26:630-4. [Crossref] [PubMed]
- Ohle R, McIsaac SM, Woo MY, et al. Sonography of the Optic Nerve Sheath Diameter for Detection of Raised Intracranial Pressure Compared to Computed Tomography: A Systematic Review and Meta-analysis. J Ultrasound Med 2015;34:1285-94. [Crossref] [PubMed]
- Robba C, Santori G, Czosnyka M, et al. Optic nerve sheath diameter measured sonographically as non-invasive estimator of intracranial pressure: a systematic review and meta-analysis. Intensive Care Med 2018;44:1284-94. [Crossref] [PubMed]
- Robba C, Cardim D, Tajsic T, et al. Non-invasive Intracranial Pressure Assessment in Brain Injured Patients Using Ultrasound-Based Methods. Acta Neurochir Suppl 2018;126:69-73. [Crossref] [PubMed]
- Hassen GW, Al-Juboori M, Koppel B, et al. Real time optic nerve sheath diameter measurement during lumbar puncture. Am J Emerg Med 2018;36:736.e1-736.e3. [Crossref] [PubMed]
- Geeraerts T, Merceron S, Benhamou D, et al. Non-invasive assessment of intracranial pressure using ocular sonography in neurocritical care patients. Intensive Care Med 2008;34:2062-7. [Crossref] [PubMed]
- Singleton J, Dagan A, Edlow JA, et al. Real-time optic nerve sheath diameter reduction measured with bedside ultrasound after therapeutic lumbar puncture in a patient with idiopathic intracranial hypertension. Am J Emerg Med 2015;33:860.e5-7. [Crossref] [PubMed]
- Dubourg J, Javouhey E, Geeraerts T. ey al. Ultrasonography of optic nerve sheath diameter for detection of raised intracranial pressure: a systematic review and meta-analysis. Intensive Care Med 2011;37:1059-68. [Crossref] [PubMed]
- Geeraerts T, Launey Y, Martin L, et al. Ultrasonography of the optic nerve sheath may be useful for detecting raised intracranial pressure after severe brain injury. Intensive Care Med 2007;33:1704-11. [Crossref] [PubMed]
- Toscano M, Spadetta G, Pulitano P, et al. Optic Nerve Sheath Diameter Ultrasound Evaluation in Intensive Care Unit: Possible Role and Clinical Aspects in Neurological Critical Patients' Daily Monitoring. Biomed Res Int 2017;2017:1621428. [Crossref] [PubMed]
- Wang L, Feng L, Yao Y, et al. Ultrasonographic Evaluation of Optic Nerve Sheath Diameter among Healthy Chinese Adults. Ultrasound Med Biol 2016;42:683-8. [Crossref] [PubMed]
- Hansen HC, Lagreze W, Krueger O, Helmke K. Dependence of the optic nerve sheath diameter on acutely applied subarachnoidal pressure - an experimental ultrasound study. Acta Ophthalmol 2011;89:e528-32. [Crossref] [PubMed]
- Launey Y, Nesseler N, Le Maguet P, et al. Effect of osmotherapy on optic nerve sheath diameter in patients with increased intracranial pressure. J Neurotrauma 2014;31:984-8. [Crossref] [PubMed]
- Dinsmore M, Han JS, Fisher JA, et al. Effects of acute controlled changes in end-tidal carbon dioxide on the diameter of the optic nerve sheath: a transorbital ultrasonographic study in healthy volunteers. Anaesthesia 2017;72:618-23. [Crossref] [PubMed]
- Killer HE, Laeng HR, Flammer J, et al. Architecture of arachnoid trabeculae, pillars, and septa in the subarachnoid space of the human optic nerve: anatomy and clinical considerations. Br J Ophthalmol 2003;87:777-81. [Crossref] [PubMed]
- Hansen HC, Helmke K. Validation of the optic nerve sheath response to changing cerebrospinal fluid pressure: ultrasound findings during intrathecal infusion tests. J Neurosurg 1997;87:34-40. [Crossref] [PubMed]
- Maissan IM, Dirven PJ, Haitsma IK, et al. Ultrasonographic measured optic nerve sheath diameter as an accurate and quick monitor for changes in intracranial pressure. J Neurosurg 2015;123:743-7. [Crossref] [PubMed]
- Chen LM, Wang LJ, Hu Y, et al. Ultrasonic measurement of optic nerve sheath diameter: a non-invasive surrogate approach for dynamic, real-time evaluation of intracranial pressure. Br J Ophthalmol 2019;103:437-41. [Crossref] [PubMed]


