
The value of high-resolution HLA in the perioperative period of non-sensitized lung transplant recipients
Introduction
Polymorphism analysis of histocompatibility leukocyte antigen (HLA) is essential for recipients in organ transplantation, which has been widely recognized to improve graft survival significantly and recipient prognosis (1,2). Especially for kidney transplantation, HLA-ABDR matching has been used as an important parameter for organ distribution in the worldwide. However, due to scarcity of lung grafts and high requirement of maintenance, currently, it is impossible to distribute donor lung according to HLA worldwide (3).
At present, most organ transplant centers use two-digit low-resolution HLA (LR-HLA) typing, also known as “Serological equivalent HLA antigen”. With the improvement of diagnostic methods, the number of alleles has increased rapidly. The practice has proved that the precise degree of HLA allele matching plays a key role and is a crucial factor affecting the long-term prognosis of transplanted organs (4). In recent years, immunologists and surgical experts have suggested that high-resolution HLA (HR-HLA) typing should be used at least in highly sensitized kidney transplant recipients (5). Recently studies (6) have shown that HLA class II eplet mismatch (MM) (DRB 1/3/4/5 + DQA/B) were poor prognosis indicators after lung transplantation, especially related to chronic lung allograft dysfunction (CLAD), contributing to the limited survival expectation of the lung transplant recipients.
In the acute phase or short-term after transplantation, eplet epitope mismatch might affect the occurrence of perioperative primary graft dysfunction (PGD) and acute rejection (AR). Thus, we conducted high-resolution HLA analysis for 59 lung transplant recipients and donors, to figure out the correspondence of HLA eplet MM and recipients’ prognosis.
Methods
Study population
We routinely conducted LR-HLA matching on all lung transplant recipients and donors from April 1, 2018, to June 30, 2019, in the center. We further conducted HR-HLA matching and Donor specific antibodies (DSA) monitoring for some of the patients considering the economic burden issue. We collected perioperative clinical data from this subset of patients with HR-HLA matching information to compare the differences between the two types of matching and the correlation between clinical PGD and AR. Donor lung distribution was automatically assigned by the National Transplant allocation system.
The Institutional Ethics Committees of Wuxi People’s Hospital afflicted to Nanjing Medical University, approved the study (No. 2018-323), including our retrospective review, verbal consent procedure, and analysis of data. All patient data were anonymous. Written informed consents were obtained from the patients or their next of kin. The research was conducted following the 2000 Declaration of Helsinki and the Declaration of Istanbul in 2008. None of the transplant donors were from a vulnerable population, and all donors or next of kin provided written informed consents that were freely given.
HLA antigen, high-resolution HLA typing, and eplet matching
In this case, HLA allelic genotyping (-A, -B, -C, -DRB1, and -DQB1/A1) was performed by sequence-based typing (SBT) based on the Luminex technology (One Lambda, Inc., Canoga Park, CA, USA). Where Luminex-based SSO results were issued, the common and well-documented (CWD) HLA alleles were listed, and in some cases, typing for HLA-DRB3/4/5 was distributed based on strong DRB1 associations. Eplet matching for all transplants was assessed by the HLA Matchmaker 500 pair (ABC and DRDQ eplet) program (http://www.hlamatchmaker.net/). All donors and recipients’ HLA typing were entered, then the program assigned each paired eplet mismatch load.
Data collection
Baseline data were composed of both recipient characteristics and transplant-related features; the former consisted of age, sex, preoperative diagnosis, and blood type; the latter included single or bilateral lung transplantation, amount of bleeding, blood transfusion, percentage of peak panel-reactive antibody (PRA), total ischemic time, type of initial immunosuppressants (categorized as tacrolimus, cyclosporin A, mycophenolate mofetil, and prednisone), time of transplant, HLA antigen mismatch, and eplet mismatch between donors and recipients.
Clinical outcomes
The primary clinical outcomes of this study were PGD (defined as the syndrome of acute lung injury early after lung transplantation), acute cellular rejection (ACR) and antibody-mediated rejection (AMR, associated with measurable allograft dysfunction) within a perioperative period (1 month). These complications can significantly influence long-term complications and survival, having a significant impact on the recipient’s quality of life and healthcare. The definition and severity grading of PGD were reported by the International Society for Heart and Lung Transplantation in 2016 (7). ACR is the consequence of an immune response of the host against the lung graft, and graft biopsy is the gold standard technique to diagnose ACR (8). The diagnosis of AMR needs at least two of the following three standards: the presence of donor-specific anti–HLA antibody, positive C4d staining on immunofluorescence, and characteristic histologic changes of AMR (9).
Statistical analyses
Continuous data were expressed as mean ± standard deviations. Comparisons between the 2 groups were performed by chi-square or Fisher test, and Student’s t-test, comparisons among three or more groups were made with one-way ANOVA analysis. Survival was estimated by Kaplan-Meier analysis. Statistical analyses were performed with the SPSS 22.0 software (SPSS Inc., USA), and P <0.05 was considered statistically significant.
Results
Study population
From April 1, 2018, to June 30, 2019, 150 cases of lung transplantation were performed in our hospital. At our center, LR-HLA matching was routinely applied to all recipients, and HR-HLA monitoring needed additional cost. Only 59 of these 150 patients received HR-HLA matching detection. There were 8 females (13.56%) and 51 males (86.44%). The average age at the time of operation was 55.73±10.78 years. The primary diseases included, 26 cases (44.07%) of interstitial lung disease (ILD) 14 cases of chronic obstructive pulmonary disease (COPD), 11 cases of pneumoconiosis, 2 cases of pulmonary hypertension (PH), 1 case of bronchiectasis, 1 case of diffuse pantothenic bronchiolitis (DPB) and 1 case of pulmonary lymphangioleiomyomatosis (PLAM). There were 31 cases of single lung transplantation and 28 cases of double lung transplantation. The average operation time was 6.09±1.48 hours. The average bleeding volume of 1,032.71±749.16 mL with blood transfusion was 852.27±901.44 mL. The average cold ischemia time was 7.65±1.93 hours (Table 1).

Full table
Immune background and immunosuppressive therapy
The ABO blood types of the donors and recipients in this group of 59 patients were completely matched. LR-HLA mismatches were 7.19±1.61, HR-HLA mismatches were 8.31±1.75 (P=0.0005). There were no high sensitization recipients in this group before the operation, of which 56 patients were PRA <10% and 3 patients were PRA 11–30%. Considering the elevated risk of perioperative infection rate, no immune induction therapy was adopted. The postoperative routine used regimen was tacrolimus (FK) + mycophenolate mofetil (MMF) + prednisone (Pred) triple immunosuppressive therapy. Among those, 50 recipients received FK + Pred dual immunosuppression treatment due to perioperative pulmonary infection; 6 patients were treated with conventional FK+MMF+ Pred triple immunosuppressive therapy. The remaining three patients were unable to tolerate the side effects of tacrolimus, so they were treated with cyclosporine (CSA) +MMF + Pred (1 case) and CSA + Pred (2 cases).
PGD occurrence
Within one week after the operation, 8 cases presented PGD 1, 15 cases of PGD 2, 32 cases of PGD 3. The relationship between PGD and HLA mismatch is shown in Table 2. Regardless of LR-HLA or HR-HLA, or HLA-DQ locus, it showed that as the increase of mismatch sites number, the severity of PGD in the early postoperative period was significantly increased (Table 2). After dividing into two groups according to PGD0-2 and 3, mismatch of HLA antigen, eplet, and HLA-DQ were positively correlated with the severity of PGD, especially eplet MM and HLA-DQ mismatch (Table 3). Also, the severity of PGD was positively correlated with mechanical ventilation time. There was no statistically significant difference between PGD and cold ischemia time, ICU time, and ECMO assisting time.
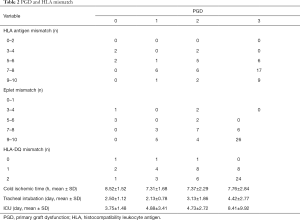
Full table

Full table
ACR occurrence
In our cohort, one patient in this group developed ACR. The patient had to be re-intubated and mechanically ventilated due to severe ACR on post-operation day 7 (POD 7), and ECMO was used. After the treatment with large doses of glucocorticoids, ECMO and mechanical ventilation were successfully weaned. The patient’s HLA antigen MM was 6, but eplet MM was as high as 9.
AMR occurrence
In this group of patients, 2 cases of AMR occurred after the operation, eplet MM 9 (HLA-DQ MM 2) and eplet MM 5 (HLA-DQ MM 1). Both patients developed DSA after operation, and they were DQB1 06:01 and C07:02, respectively.
Death
There were 9 cases of death during the perioperative period. Five of them died of severe PGD and 4 died of severe infection. The 9 patients’ eplet MMs were at average of 9.4, HLA-DQ MMs were at an average of 1.9.
Discussion
The status of HLA in organ transplantation has been widely recognized. HLA matching improved organ transplantation results and benefited recipients (10,11). HLA matching supplied benefits in improving outcomes in kidney transplantation and remained part of the kidney allocation. HLA-ABDR was included in the allocation, but HLA-DQ was not considered (12-14). However, due to the short cold ischemia time requirement of donated lungs, it has not been possible to allocate donors according to the degree of HLA matching in lung transplantation in the past. HLA typing has been more used in retrospective analysis at the lung transplant center to guide later immunosuppressive therapy.
Although HLA-DQ and -DR may be closely related at an antigen level, it has been shown that small differences in one or more epitopes between donors and recipients at either locus were sufficient to generate a humoral and/or T cell-mediated immune response (15,16). In 2015, Hahn reported that a 41-year-old female patient received a 0 MM kidney and pancreas in 1998 and 2000, respectively. However, when she planned to undergo a second kidney transplant in 2014, PRA level was as high as 84%, while an antibody that conflicted with its HLA-A2 site existed. This case report illustrated the importance of high-resolution HLA typing, suggesting that low-resolution HLA typing was problematic and unreliable (17). Huang et al. retrospectively analyzed the data of HR-2F HLA in solid organ transplantation applications at Children's Hospital of Philadelphia and Temple Hospital, a better result could be seen when HLA typing was performed at the HR-2F level (18). Our data showed that this group of patients had LR-HLA mismatch 7.19±1.61, eplet mismatch 8.31±1.75, showing that there was a significant difference between the two methods’ presentation. When analyzing the relationship between the two matching methods and clinical PGD manifestation, there was still statistically discrepancy. We further need to evaluate the joint results of organ acquisition, transit time, pulmonary artery pressure, blood loss and other factors related to surgical operations and treatments.
A recent “Personal Viewpoint” paper addressed the concept that HLA typing at the four-digit or allele level offered a more exact approach to find suitable donors for sensitized patients (5). Our recipients were non-sensitized, and the results showed that eplet matching was closely related to perioperative PGD. This suggested the importance of precise matching in lung transplantation. Huang et al. conducted HR-2F HLA typing results showed that the most frequent use of HR-2F HLA typing was for postoperative monitoring of DSA. As in our study, 2 patients had AMR with DSA. Without HR-HLA data of donor and recipient, it would be hard for determination and prediction. However, our results were quite preliminary, and further work should be done to investigate the relationship of the HLA MM and clinical prognosis.
In 2016, Lim et al. published a median follow-up of 2.8 years for 788 recipients of kidney transplantation in Australia. Among these patients, 321 (40.7%) patients were with HLA-DQ 0 MM, 467 (59.4%) with 1–2 MM (19). The research showed an independent association between HLA-DQ mismatches and acute rejection, including AMR. It is important to point out that most of the acute rejection (80%) occurred within the first 6 months after transplantation, suggesting the potential contribution of pre-transplant donor-specific anti-HLA-DQ antibody to the risk of early rejection. Therefore, the authors suggested that the degree of HLA-DQ site matching should be added to the current deceased donor kidney distribution system (20,21). HLA-DQ mismatching was associated with lower graft survival independent of HLA-ABDR in living donor kidney transplants and deceased donor kidney transplants, with a higher 1-year risk of acute rejection (22). In acute graft versus host disease after hematopoietic stem cell transplantation, donor-recipient incompatibility at the HLA-DQ locus was associated with a two-fold greater risk of acute graft-versus-host disease, independent of compatibility at the HLA-DR locus (23,24). Accordingly, once the recipient had a de novo DSA to HLA-DQ, the risk of AMR increased as to 10-fold, which was often associated with early graft loss (25-27). This study found that HLA-DQ MM was strongly associated with severe PGD after lung transplantation. At the same time, we also observed a total of 9 patients died in this group, 5 died of severe PGD, 4 died of severe infection, especially that they all had elevated levels of eplet MMs and HLA-DQ MMs.
In conclusion, perioperative PGD and long-term CLAD were the most detrimental results with managing difficulties in lung transplantation. Pre-detection of eplet matching and DSA could accurately reflect the genetic background of donors and recipients; thus predicting the risk of early PGD and acute rejection after lung transplantation. Donations can be made more effective if the organ distribution can be further guided by HLA eplet matching in lung transplantation. Thus, the allografts can survive even longer with postoperative complications and immunosuppressive strength to be reduced.
Acknowledgments
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The Institutional Ethics Committees of Wuxi People’s Hospital afflicted to Nanjing Medical University, approved the study (No. 2018-323), including our retrospective review, verbal consent procedure, and analysis of data. All patient data were anonymous. Written informed consents were obtained from the patients or their next of kin.
References
- Hart A, Smith JM, Skeans MA, et al. OPTN/SRTR 2016 Annual Data Report: Kidney. Am J Transplant 2018;18 Suppl 1:18-113. [Crossref] [PubMed]
- Hricik DE. Primer on transplantation. 3rd ed. 2011; Chichester, West Sussex; Hoboken, N.J.: Wiley-Blackwell. ix, 307 p.
- Ju L, Suberbielle C, Li X, et al. HLA and lung transplantation. Front Med 2019;13:298-313. [Crossref] [PubMed]
- Flomenberg N, Baxter-Lowe LA, Confer D, et al. Impact of HLA class I and class II high-resolution matching on outcomes of unrelated donor bone marrow transplantation: HLA-C mismatching is associated with a strong adverse effect on transplantation outcome. Blood 2004;104:1923-30. [Crossref] [PubMed]
- Duquesnoy RJ, Kamoun M, Baxter-Lowe LA, et al. Should HLA mismatch acceptability for sensitized transplant candidates be determined at the high-resolution rather than the antigen level. Am J Transplant 2015;15:923-30. [Crossref] [PubMed]
- Walton DC, Hiho SJ, Cantwell LS, et al. HLA Matching at the Eplet Level Protects Against Chronic Lung Allograft Dysfunction. Am J Transplant 2016;16:2695-703. [Crossref] [PubMed]
- Snell GI, Yusen RD, Weill D, et al. Report of the ISHLT Working Group on Primary Lung Graft Dysfunction, part I: Definition and grading-A 2016 Consensus Group statement of the International Society for Heart and Lung Transplantation. J Heart Lung Transplant 2017;36:1097-103. [Crossref] [PubMed]
- Stewart S, Fishbein MC, Snell GI, et al. Revision of the 1996 working formulation for the standardization of nomenclature in the diagnosis of lung rejection. J Heart Lung Transplant 2007;26:1229-42. [Crossref] [PubMed]
- Levine DJ, Glanville AR, Aboyoun C, et al. Antibody-mediated rejection of the lung: A consensus report of the International Society for Heart and Lung Transplantation. J Heart Lung Transplant 2016;35:397-406. [Crossref] [PubMed]
- Gilks WR, Bradley BA, Gore SM, et al. Substantial benefits of tissue matching in renal transplantation. Transplantation 1987;43:669-74. [Crossref] [PubMed]
- Doxiadis II, de Fijter JW, Mallat MJ, et al. Simpler and equitable allocation of kidneys from postmortem donors primarily based on full HLA-DR compatibility. Transplantation 2007;83:1207-13. [Crossref] [PubMed]
- Wissing KM, Fomegné G, Broeders N, et al. HLA mismatches remain risk factors for acute kidney allograft rejection in patients receiving quadruple immunosuppression with anti-interleukin-2 receptor antibodies. Transplantation 2008;85:411-6. [Crossref] [PubMed]
- Beckingham IJ, Dennis MJ, Bishop MC, et al. Effect of human leucocyte antigen matching on the incidence of acute rejection in renal transplantation. Br J Surg 1994;81:574-7. [Crossref] [PubMed]
- Lim WH, Chadban SJ, Clayton P, et al. Human leukocyte antigen mismatches associated with increased risk of rejection, graft failure, and death independent of initial immunosuppression in renal transplant recipients. Clin Transplant 2012;26:E428-37. [Crossref] [PubMed]
- Duquesnoy RJ. A structurally based approach to determine HLA compatibility at the humoral immune level. Hum Immunol 2006;67:847-62. [Crossref] [PubMed]
- Dankers MK, Witvliet MD, Roelen DL, et al. The number of amino acid triplet differences between patient and donor is predictive for the antibody reactivity against mismatched human leukocyte antigens. Transplantation 2004;77:1236-9. [Crossref] [PubMed]
- Hahn AB, Bravo-Egana V, Jackstadt JL, et al. HLA-A2 reactive antibodies in a patient who types as HLA-A2: The importance of high resolution typing and epitope-based antibody analysis. Transpl Immunol 2015;32:141-3. [Crossref] [PubMed]
- Huang Y, Dinh A, Heron S, et al. Assessing the utilization of high-resolution 2-field HLA typing in solid organ transplantation. Am J Transplant 2019;19:1955-63. [Crossref] [PubMed]
- Lim WH, Chapman JR, Coates PT, et al. HLA-DQ Mismatches and Rejection in Kidney Transplant Recipients. Clin J Am Soc Nephrol 2016;11:875-83. [Crossref] [PubMed]
- Wiebe C, Pochinco D, Blydt-Hansen TD, et al. Class II HLA epitope matching-A strategy to minimize de novo donor-specific antibody development and improve outcomes. Am J Transplant 2013;13:3114-22. [Crossref] [PubMed]
- Sapir-Pichhadze R, Tinckam K, Quach K, et al. HLA-DR and -DQ eplet mismatches and transplant glomerulopathy: a nested case-control study. Am J Transplant 2015;15:137-48. [Crossref] [PubMed]
- Leeaphorn N, Pena JRA, Thamcharoen N, et al. HLA-DQ Mismatching and Kidney Transplant Outcomes. Clin J Am Soc Nephrol 2018;13:763-71. [Crossref] [PubMed]
- Petersdorf EW, Longton GM, Anasetti C, et al. Definition of HLA-DQ as a transplantation antigen. Proc Natl Acad Sci U S A 1996;93:15358-63. [Crossref] [PubMed]
- Fernández-Viña MA, Klein JP, Haagenson M, et al. Multiple mismatches at the low expression HLA loci DP, DQ, and DRB3/4/5 associate with adverse outcomes in hematopoietic stem cell transplantation. Blood 2013;121:4603-10. [Crossref] [PubMed]
- Willicombe M, Brookes P, Sergeant R, et al. De novo DQ donor-specific antibodies are associated with a significant risk of antibody-mediated rejection and transplant glomerulopathy. Transplantation 2012;94:172-7. [Crossref] [PubMed]
- Ginevri F, Nocera A, Comoli P, et al. Posttransplant de novo donor-specific hla antibodies identify pediatric kidney recipients at risk for late antibody-mediated rejection. Am J Transplant 2012;12:3355-62. [Crossref] [PubMed]
- Devos JM, Gaber AO, Teeter LD, et al. Intermediate-term graft loss after renal transplantation is associated with both donor-specific antibody and acute rejection. Transplantation 2014;97:534-40. [Crossref] [PubMed]


