
Mitral valve replacement via minimally invasive totally thoracoscopic surgery versus traditional median sternotomy: a propensity score matched comparative study
Introduction
Cardiac surgery via median sternotomy (MS) as a conventional approach has its drawbacks including inevitable blood loss and transfusion, unbearable postoperative pain, and a long period of time for recovery (1). In order to improve postoperative outcomes, minimally invasive approaches including via an upper and lower incision on the sternum and via left and right anterolateral incisions have been performed (2,3). Compared to conventional MS, these minimally invasive approaches reduce trauma to the sternum but still require exposure of the ascending aorta and the superior and inferior vena cava (IVC) to establish the central cardiopulmonary bypass (CPB), which limits further minimization of invasiveness in cardiac surgery.
With the innovation of the port access approach in the late 1990s, the success of the first thoracoscopic-assisted minimally invasive mitral valve (MV) surgery via a small right anterolateral incision had shown that minimally invasive thoracoscopic cardiac surgery with peripheral CPB was as feasible, safe, and effective as the conventional MS approach (2,4-8). However, performing a thoracoscopic cardiac surgery usually meant the surgeon was limited to a very narrow surgical field with poor exposure and required the use of long-shafted endoscopic instruments, which markedly increased the learning difficulty and learning curve (9,10). Furthermore, early research showed that the CPB time and aortic cross-clamping time in minimally invasive thoracoscopic cardiac surgery was significantly longer than that in MS, and the risk of stroke was higher in the minimally invasive group (11-13).
In this study, propensity score matching (PSM) was used to enable proper comparative retrospective analysis of the data from patients who underwent MV replacement (MVR) surgery via minimally invasive thoracoscopy versus those who underwent traditional MS, to determine and compare the short-term mortality and long-term post cardiac surgery outcomes of these two approaches. This study was aimed to evaluate the safety and efficacy of thoracoscopic cardiac surgery and provide guidance for surgeons who intend to perform thoracoscopic cardiac surgery in China.
Methods
Patients
From January 1st, 2012 to June 30th, 2015, a total of 1,096 patients who underwent MVR surgery concomitant with tricuspid valve surgery or atrial fibrillation ablation therapy in the Department of Cardiovascular Surgery of Guangdong Cardiovascular Institute were analyzed. Patients undergoing other concomitant procedures such as aortic surgery, aortic valve replacement, or coronary artery bypass graft were excluded. Patients who were younger than 18 years old, or undergoing a second MVR or conversion to MVR from a mitral valvuloplasty procedure were also excluded. Emergency surgery cases were also excluded from this study. Of the 1,096 patients, 405 were operated on with a minimally invasive surgery of the MVR (MIs-MVR), and 691 cases were operated on with MS-MVR.
The primary outcome was a composite end point consisting of all important adverse events, including 30-day mortality, stroke, aortic dissection, perioperative myocardial infarction, new atrial fibrillation, intraoperative conversion to sternotomy, re-exploration for bleeding, and low-cardiac-output syndrome with necessity for intra-aortic balloon pump. Follow-up vital status was obtained by telephone or routine clinic visit. The 36-item Short Form Health Survey was used for the last follow-up.
Surgical technique
Minimally invasive thoracoscopic MVR surgery
CPB was instituted via femoral arterial and venous cannulation through a 2–3 cm transverse incision in the right groin. Retrograde perfusion was performed through the right femoral artery (18–24 Fr). The tip of the venous cannula was positioned in the IVC (22–28 Fr), and then a second venous cannula was inserted percutaneously through the right internal jugular vein and positioned in the superior vena cava (SVC) (16–18 Fr). Patient temperature was cooled to 34 °C, and vacuum-assisted CPB was used throughout the procedure.
The surgical approach was performed via two ports in the right chest (Figure 1).
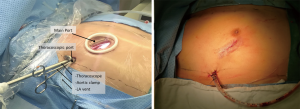
Main port
A right lateral mini-thoracotomy, about 3.5–4.0 cm in length, was performed in the 4th intercostal space; the specific size should be sufficient for the artificial valve to be passed through. For male patients, the incision was placed just below and lateral to the nipple, and for female patients, the incision was placed in the sub-mammary crease. A small tissue retractor was utilized to protect the incision.
Thoracoscopic port
A video camera was inserted through a 10-mm port in the 4th intercostal space in the proximal midaxillary line. A transthoracic Chitwood aortic cross-clamp and a left ventricular (LV) vent were also inserted through this port.
Antegrade crystalloid Bretschneider cardioplegia (2:l) was administered directly into the aortic root, and then continued for 90–120 min if necessary. The surgical field was flooded with CO2 through the camera port throughout the procedure. The pleura should be opened after the patient is placed on CPB. The left atrium was opened posterior to the interatrial groove. A left atrial retractor was used to expose the MV. Specialized long-shafted surgical tools were utilized for tissue handling and suturing. Standard MV replacement was performed under totally endoscopic vision. Concomitant tricuspid valvuloplasty could be performed by blocking the SVC and the LV compliance (LVC) through the right atrium. Deairing was performed via a left ventricle drainage tube and the cardioplegia puncture site on the ascending aorta.
Intraoperative transesophageal echocardiography was used to determine the immediate results of the replacement and check for perivalvular leakage and residual bubble. If the outcome was satisfactory and there was no active bleeding at the incisions, the pericardium was closed with interrupted sutures, and the incisions were sutured using the thoracoscope. CPB should be stopped after the central temperature returns to normal. Two-lung ventilation was then conducted, and a thoracic drainage tube was placed through the camera port.
Traditional MS-MVR
A standard sternal median incision was performed, generally about 22–26 cm in length from a point 1–2 cm below the fossa suprasternalis to the xiphoid. A sternal retractor was used to expose the mediastinum, and the pericardium was opened to expose the heart under direct vision. CPB was established by ascending aortic cannulation, and superior and IVC cannulation. A drainage tube was placed in the left superior pulmonary vein for a better surgical field. Myocardial protection was performed as above, and ice water was poured into the pericardium. Standard MVR was performed through the interatrial groove, and concomitant tricuspid valvuloplasty was performed as necessary. Two drainage tubes were placed in the pericardium and mediastinum before the sternum was closed.
Statistical analysis
In this study, PSM was used to reduce the effect of potential confounding factors in observational study. The PSM was performed by matching patients in the two groups at a 1:1 ratio, without replacement, by the nearest neighbor technique. PSM was performed with age, sex, LV ejection fraction (LVEF), pulmonary artery pressure, Euroscore II value, type of lesion, and surgical technique as the observation indicators, and using SPSS 21.0 software (IBM, Armonk, NY, USA). This resulted in 404 matched cases: 202 in the MIs group, and 202 in the MS group. The criterion for matching pairs used a caliper width equal to 0.2 of the pooled SD of the logit of propensity score. After matching, there was no significant difference between the two groups in New York Heart Association (NYHA) cardiac function classification, valvular lesion type, or surgical technique (all P>0.05, Table 1).
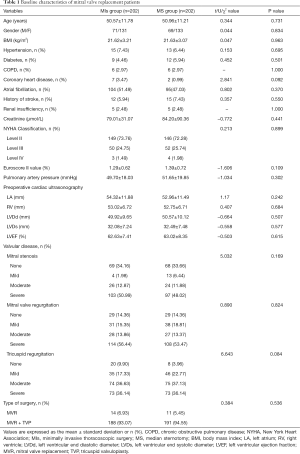
Full table
Normal distributed continuous variables are expressed as the mean ± standard deviation, and categorical variables as proportions throughout this study. Continuous variables were analyzed using the Student’s t-test or the Mann-Whitney U test of two independent samples. Categorical variables were analyzed using the χ2 test. Survival curves were analyzed with Kaplan-Meier actuarial methods, and log-rank was used to detect the differences between groups. All tests were two-tailed, and P values <0.05 were considered significant. SPSS 21.0 software was used for the statistical analyses, and the follow-up data were recorded using EpiData software (Version 3.1, Odense, Denmark, 2004).
Results
MVR surgery was successfully completed in the two matched groups of patients, with no cases of surgical failure. There was no intraoperative conversion to median thoracotomy surgery in the MIs group. Compared with the MS group, the MIs group had significantly longer CPB time, aortic cross-clamping time, and operation time (P<0.001), but a significantly lower intraoperative transfusion rate and fewer transfusion units (both P<0.05, Table 2).
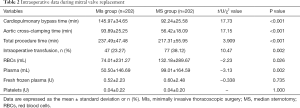
Full table
The early postoperative data showed that the MS group, when compared with the MIs group, had a significantly longer length of ventilation and ICU stay, significantly increased chest tube drainage, significantly increased postoperative transfusion rate and volume, significantly longer duration of chest tube drainage and postoperative hospital stay, and significantly increased total hospitalization cost (all P<0.05, Table 3). However, there was no significant difference between groups in the need for re-exploration for bleeding and postoperative LVEF. There was no significant difference between the two groups in the morbidity rate of in-hospital death, stroke, pulmonary infection, arrhythmia, acute renal failure, or chylothorax, but the MS group had a significantly increased incision infection rate (P=0.004), and a significantly higher morbidity rate of all the early postoperative complications (P=0.038, Tables 3,4).
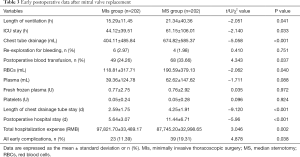
Full table

Full table
In this study, 8 patients died during early postoperative hospitalization (3 in the MIs group and 5 in the MS group). Cause of death in the MIs group was stroke, lung infection, and disseminated intravascular coagulation (n=2); and pulmonary infection and malignant arrhythmia (n=1). In the MS group, cause of death was stroke, pulmonary infection and multiple organ failure (n=2); low cardiac output syndrome and malignant cardiac arrhythmia (n=1); and pulmonary infection and renal failure (n=2).
Telephone or outpatient follow-up was conducted for a total of 396 patients (199 in the MIs group, and 197 in the MS group) who were discharged from the hospital 6–48 months postoperatively, with an average follow-up period of 26.59±12.33 months. There was a total of 361 valid follow-ups, with 35 patients being unable to be followed up due to change of contact information (16 in the MIs group and 19 in the MS group); the withdrawal rate was 8.84%.
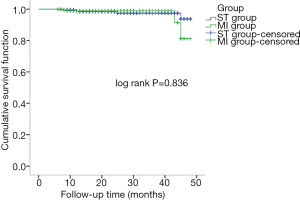
Nine patients (2.27%) died during follow-up. In the MIs group, 2 patients died of stroke and pulmonary infection, 1 died of malignant cardiac arrhythmia, and 1 died of unknown causes. In the MS group, 2 patients died from stroke, 2 died from renal failure and pulmonary infection, and there was 1 case of sudden death for unknown reasons. A comparison of Kaplan-Meier survival curves (Figure 2) and log-rank testing showed no significant difference in survival between the two groups (P=0.836). There was no significant difference between the two groups in the rate of reoperation, the need to install a permanent pacemaker, or the incidence of stroke (Table 5). Postoperative mediastinal infection was significantly higher in the MS group than in the MIs group (P=0.028, Table 5). There was no significant difference between the two groups in terms of postoperative ability to perform self-care, the ability to resume work or study, or NYHA classification (Table 6). However, in the early postoperative period, the MIs group took a significantly shorter length of time to resume self-care and work or study than the MS group (all P<0.05, Table 6). Long-term follow-up found that there was a significantly greater proportion of patients in the MIs group who were satisfied with the incision compared with the MS group (P<0.001, Table 6). There was no significant difference between the two groups in all items of the 36-item Short Form Health Survey (SF-36) (Table 7).

Full table

Full table

Full table
Discussion
In the past few decades, minimally invasive methods of cardiac surgery have continuously developed and improved, with thoracoscopic cardiac surgery technology advancing accordingly (14). Traditional MS cardiac surgery now provides good surgical vision and outcomes (15-17); however, it requires that the sternum be completely sawed through which damages sternal integrity, increases bleeding and postoperative pain, can potentially cause the creation of a hernia under the xiphisternum or mediastinal infection, and leaves residual permanent steel wire (15-17). Cardiac surgery via MIs could potentially have better outcomes than traditional MS; however, it is still unclear which of the two approaches is superior. MIs requires specialized long-shafted instruments, and visualization via video-assisted thoracoscopy is different from traditional approaches. It is also unclear whether the prolonged CPB and aortic cross-clamping time in thoracoscopic surgery would increase the short- and long-term mortality, stroke, and other complications (13). Hence, further research is warranted to determine the safety and efficacy of minimally invasive thoracoscopic cardiac surgery.
A prospective controlled study of 40 cases of cardiac surgery via thoracoscopy versus MS found no significant difference between the two groups in surgical mortality (18). Similarly, in the present study, although the MIs group had significantly longer CPB time, aortic cross-clamping time, and total procedure time than the MS group, there was no significant difference between the two groups regarding in-hospital mortality rate.
Whether thoracoscopic cardiac surgery increases the risk of stroke and other complications compared with conventional sternotomy remains controversial. In order to facilitate a better venous drainage and a clear surgical field, thoracoscopic cardiac surgery requires cannulations of both the right internal jugular vein and the femoral vein along with single-lung ventilation during procedure. Cheng et al. (12) reported that minimally invasive MV surgery significantly increased the postoperative stroke rate. However, a meta-analysis of 14,390 cases of cerebral embolism after minimally invasive or median thoracotomy MV surgery showed that there was no significant difference between the groups in the rate of cerebral embolism (19). In order to prevent perioperative stroke in the study, the following measures were used during thoracoscopic cardiac surgery: (I) high flow of CO2 administered before incising the left atrium, and a continuous low flow of CO2 used in the chest intraoperatively; (II) active sucking of the aorta via cardioplegia puncture before clamp-off and no residual gas confirmed by transesophageal echocardiography before weaning of CPB; (III) preoperative computed tomography angiography of the chest and abdominal blood vessels conducted in patients aged >70 years.
In this study, MVR was successfully completed in all cases in the MIs group, and there was no significant difference between the two groups in stroke, lung infection, and other complications. Moreover, the morbidity of all postoperative complications was significantly lower in the MIs group compared to the MS group.
Minimally invasive thoracoscopic cardiac surgery may potentially have benefits over traditional thoracotomy. Thoracoscopic cardiac surgery is conducted through a small incision on the right chest, which does not damage the median sternum and thus greatly reduces the degree of surgical trauma compared with conventional sternotomy. The intercostal port and tissue retractor markedly reduce the damage and pain caused to the surrounding muscle tissues (20). The applications of a high-resolution graphic system and 10 mm camera lens enable a more precise and clearer surgical vision, which provides better observation of intraoperative bleeding and allows more accurate operation. Therefore, it reduces transfusion rate and transfusion-related complications, postoperative chest tube drainage, and length of chest tube drainage stay (18,19). In this study, the MIs group had significantly less chest tube drainage, shorter stay of chest tube drainage, a lower rate of intra- and postoperative transfusion, and a lower transfusion volume than the MS group.
Minimally invasive thoracoscopic cardiac surgery improves early postoperative recovery compared with MS. Thoracoscopic cardiac surgery maintains the integrity and stability of the thorax and decreases the early postoperative incision pain compared with the MS approach. Minimally invasive thoracoscopic cardiac surgery is also conducive to early postoperative respiratory function recovery and shortens the ventilation time and ICU stay. In addition, earlier postoperative off-bed activity is conducive to gastrointestinal motility and reduces the incidence of postoperative complications related to long-term rest in bed (12,19,20). Wang et al. (20) reported that patients who underwent thoracoscopic surgery had significantly shorter ventilation time compared with the MS group, and that the duration of postoperative recovery until normal activity was 6 weeks after thoracoscopic surgery versus 10 weeks after MS. Similarly, Iribarne et al. (19) reported that thoracoscopic patients resumed postoperative standing and walking after a significantly shorter period than the MS group. In this study, the length of ventilation, ICU stay, and postoperative hospital stay in the MIs group were significantly shorter than those in the MS group. Postoperatively, most patients in both groups resumed self-care and had improved heart function, but the time taken for MIs patients to resume self-care, work, or study was significantly shorter compared with MS patients, and the rate of resumption of work or study reached up to 60% at 6 weeks postoperatively. Hence, thoracoscopic cardiac surgery reduced medical costs and provided social and economic benefits.
To obtain a more objective assessment of the long-term effects of the two surgical methods, 352 patients in the present study completed the SF-36. The SF-36 results showed no significant differences between the two groups in physical and psychological status.
Serious complications after thoracoscopic cardiac surgery reported no difference to those found after median thoracotomy. Iribarne et al. (21) reported a study of 764 follow-up patients who underwent cardiac surgery via minimally invasive surgery versus MS and found no significant difference between the two groups in short- and long-term survival, with a mean survival of more than 85% after 4 years. Cheng et al. (12) reported a low morbidity of postoperative mediastinal infection for patients who underwent MIs. In this study, over an average follow-up period of 6–48 months, the 1-year survival rate was 98.86% (348/352), and the overall survival rate was 97.44% (343/352); there was no significant difference between the two groups in survival. There was also no significant difference between the two groups in rates of mortality, reoperation, stroke, or permanent pacemaker implantation; however, the MS group had a significantly higher rate of mediastinal infection.
In the early postoperative period, the use of a thoracoscopic port reduced the incidence of poor wound healing, reduced scarring, and increased the concealment of the incision. Casselman et al. (22) investigated 187 patients who underwent minimally invasive MV surgery and found that 98% of patients were satisfied with the cosmetic outcome of the surgical incision. Among the 352 patients surveyed in this study, the incidence of early poor incision healing was significantly lower in the MIs group, and significantly more MIs patients were very satisfied with the appearance of the incision compared with the MS patients.
In conclusion, minimally invasive thoracoscopic cardiac surgery does not significantly increase mortality rate, stroke, or other complications in the short- and long-term, and there is no difference in the long-term cardiac function or quality of life compared with the traditional MS approach. In addition, thoracoscopic cardiac surgery results in minimal trauma, quick recovery, less blood transfusion, and superior aesthetic outcome. Thoracoscopic cardiac surgery is safe, effective, and feasible compared with traditional MS.
Acknowledgments
Funding: This research was supported by the Grant of Guangdong Provincial People’s Hospital (2017zh06), the Guangdong Project of Science and Technology (2017B090904034), the Nature Science Foundation of Guangdong Province (2016A030313799), the Medical Research Foundation of Guangdong Province (A2016367).
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: This retrospective study was approved by the Institutional Review Board (IRB) of Guangdong Provincial People’s Hospital [approval ID: No. GDREC2016054H(R1)]. All patients signed the informed consents before surgery. The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
References
- Shibata T, Kato Y, Motoki M, et al. Mitral valve repair with loop technique via median sternotomy in 180 patients. Eur J Cardiothorac Surg 2015;47:491-6. [Crossref] [PubMed]
- Saunders PC, Grossi EA, Sharony R, et al. Minimally invasive technology for mitral valve surgery via left thoracotomy: experience with forty cases. J Thorac Cardiovasc Surg 2004;127:1026-31. [Crossref] [PubMed]
- Doty DB, Flores JH, Doty JR. Cardiac valve operations using a partial sternotomy (lower half) technique. J Card Surg 2000;15:35-42. [Crossref] [PubMed]
- Fromes Y, Gaillard D, Ponzio O, et al. Reduction of the inflammatory response following coronary bypass grafting with total minimal extracorporeal circulation. Eur J Cardiothorac Surg 2002;22:527-33. [Crossref] [PubMed]
- Carpentier A, Loulmet D, Carpentier A, et al. Open heart operation under videosurgery and minithoracotomy. First case (mitral valvuloplasty) operated with success. C R Acad Sci III 1996;319:219-23. [PubMed]
- McClure RS, Cohn LH, Wiegerinck E, et al. Early and late outcomes in minimally invasive mitral valve repair: an eleven-year experience in 707 patients. J Thorac Cardiovasc Surg 2009;137:70-5. [Crossref] [PubMed]
- Vallabhajosyula P, Wallen TJ, Solometo LP, et al. Minimally invasive mitral valve surgery utilizing heart port technology. J Card Surg 2014;29:343-8. [Crossref] [PubMed]
- Holzhey DM, Seeburger J, Misfeld M, et al. Learning minimally invasive mitral valve surgery: a cumulative sum sequential probability analysis of 3895 operations from a single high-volume center. Circulation 2013;128:483-91. [Crossref] [PubMed]
- Murzi M, Miceli A, Cerillo AG, et al. Training surgeons in minimally invasive mitral valve repair: a single institution experience. Ann Thorac Surg 2014;98:884-9. [Crossref] [PubMed]
- Modi P, Hassan A, Chitwood WJ. Minimally invasive mitral valve surgery: a systematic review and meta-analysis. Eur J Cardiothorac Surg. 2008;34:943-52. [Crossref] [PubMed]
- Mohr FW, Falk V, Diegeler A, et al. Minimally invasive port-access mitral valve surgery. J Thorac Cardiovasc Surg 1998;115:567-74, 574-6.
- Cheng DC, Martin J, Lal A, et al. Minimally invasive versus conventional open mitral valve surgery: a meta-analysis and systematic review. Innovations (Phila) 2011;6:84-103. [Crossref] [PubMed]
- Grossi EA, Loulmet DF, Schwartz CF, et al. Evolution of operative techniques and perfusion strategies for minimally invasive mitral valve repair. J Thorac Cardiovasc Surg 2012;143:S68-70. [Crossref] [PubMed]
- Bek EL, Yun KL, Kochamba GS, et al. Effective median sternotomy closure in high-risk open heart patients. Ann Thorac Surg 2010;89:1317-8. [Crossref] [PubMed]
- Plass A, Grunenfelder J, Reuthebuch O, et al. New transverse plate fixation system for complicated sternal wound infection after median sternotomy. Ann Thorac Surg 2007;83:1210-2. [Crossref] [PubMed]
- Levisman J, Shemin RJ, Robertson JM, et al. Migrated sternal wire into the right ventricle: case report in cardiothoracic surgery. J Card Surg 2010;25:161-2. [Crossref] [PubMed]
- Dogan S, Aybek T, Risteski PS, et al. Minimally invasive port access versus conventional mitral valve surgery: prospective randomized study. Ann Thorac Surg 2005;79:492-8. [Crossref] [PubMed]
- Sündermann SH, Sromicki J, Rodriguez Cetina Biefer H, et al. Mitral valve surgery: right lateral minithoracotomy or sternotomy? A systematic review and meta-analysis. J Thorac Cardiovasc Surg 2014;148:1989-1995.e4. [Crossref] [PubMed]
- Iribarne A, Easterwood R, Russo MJ, et al. Comparative effectiveness of minimally invasive versus traditional sternotomy mitral valve surgery in elderly patients. J Thorac Cardiovasc Surg 2012;143:S86-90. [Crossref] [PubMed]
- Wang D, Wang Q, Yang X, et al. Mitral valve replacement through a minimal right vertical infra-axillary thoracotomy versus standard median sternotomy. Ann Thorac Surg 2009;87:704-8. [Crossref] [PubMed]
- Iribarne A, Russo MJ, Easterwood R, et al. Minimally invasive versus sternotomy approach for mitral valve surgery: a propensity analysis. Ann Thorac Surg 2010;90:1471-7. [Crossref] [PubMed]
- Casselman FP, Van Slycke S, Dom H, et al. Endoscopic mitral valve repair: feasible, reproducible, and durable. J Thorac Cardiovasc Surg 2003;125:273-82. [Crossref] [PubMed]


