
Changes to the American Joint Committee on Cancer staging system for spine tumors—practice update
Introduction
Cancer staging systems were initially developed in the 1950s under the auspices of the Union for International Cancer Control and the American Joint Committee on Cancer (AJCC). Regular updates to the staging manual have been published since the 1970s at 6–8-year intervals. The 8th edition staging manual was recently published and applies to all cases diagnosed since January 1, 2018 (1).
The goal of a cancer staging system is to stratify patients into different categories to represent a logical progression of disease. Staging allows patients and clinicians to identify prognosis, to select among different treatments, and to evaluate patient and treatment outcomes in a standardized way to account for different disease presentations. The Enneking staging system is a well-recognized staging system in musculoskeletal oncology (2). However, it is designed for use with extremity rather than spine tumors. The Weinstein-Boriani-Biagini (WBB) system is also well-recognized in spine tumor surgery (3). However, the WBB system is a surgical (rather than oncologic) staging system designed to define the local extent of a tumor; it does not specify tumor grade or the presence or absence of distant spread. As such, the WBB system defines local tumor anatomy but does not fulfill the goals of an oncologic staging system. The 8th edition staging manual presented here is the current standard oncologic staging system for spine tumors cared for at all American cancer centers and is adopted by many other systems around the world.
Traditional staging criteria focused on anatomic criteria (tumor size, presence of lymph node or distant metastases) reflected in the TNM classification familiar to physicians. Recent updates have begun to incorporate non-anatomic criteria into the staging of some tumors [for example, prostate-specific antigen (PSA) levels in prostate cancer]. The 8th edition staging formulation for bone tumors (covering all primary malignant tumors of bone except lymphoma or myeloma) now incorporates skeletal location in addition to traditional anatomic criteria.
Rationale for change
The goal of a staging system is to reliably separate patients into different prognostic groups to aid clinicians and researchers. The relatively poor prognosis of spine tumors has been recognized in many studies, but almost all were admittedly small and generally retrospective (4-7). This recognition prompted an analysis of 5,671 bone tumor patients prospectively collected between 2002–2008 as a part of the National Cancer Data Bank collection system. This was collected using the AJCC 6th edition staging manual in place at that time, although little changes had been made to bone tumor staging in recent iterations.
The 6th edition AJCC bone staging system provides good discrimination of patient outcomes for extremity sarcomas. For example, analyzing patient outcome by stage for extremity osteosarcoma shows reliable discrimination of patient outcome by current stage groupings (Figure 1). However, a similar analysis of patient outcome for spine osteosarcomas (Figure 2) is notable in two ways. First, standard stage groupings fail to discriminate between different patient outcomes in the way that these groupings did for extremity osteosarcoma. Second, the overall prognosis of patients with spinal osteosarcoma is inferior compared to that of extremity tumors.
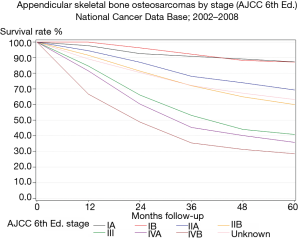
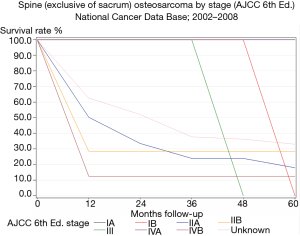
The reason for a differential prognosis based on anatomic location is unclear. These results are stratified for tumor size, grade, and presence of metastases (the traditional known risk factors for outcome in bone tumors) (8,9). There is no known difference in biologic aggressiveness of osteosarcoma based on anatomic location; in fact, spine sarcomas are often smaller in tumor volume than extremity sarcomas because of the relatively early appearance of neurologic symptoms. However, it is clear that traditional staging criteria fail to discriminate patient outcomes in spine tumors. Similar results are seen for other spine sarcomas (1).
Surgical margin is known to be a key determinant of local recurrence and patient outcome in bone tumor treatment (10-16). Spine tumors provide an anatomic challenge in reliably obtaining an oncologic margin. Absent catastrophic neurologic sacrifice, a true wide resection is rarely obtainable in the spine; as well, even with deliberate neurologic sacrifice, a wide margin may still be unobtainable in areas of retroperitoneal extension. A marginal resection with no tumor violation may be obtained for some tumors. However, many spine tumors present with an adverse local configuration which makes even a marginal resection without tumor violation difficult or impossible to reliably obtain. Unlike extremity tumors with adverse presentations, amputation is not an anatomic option. Rare cases of spinal cord resection have been reported (and the author has experience with this as well) (17). However, the potential increase in cure must be balanced again the very real health impacts from resulting paraplegia. En bloc resection including resection of the spinal cord is generally reserved for patients in whom a fixed, near complete neurologic deficit is present with little hope of functional recovery.
Readers should realize that the oncologic literature on spine tumors often suffers from poor definition of terms. For example, vertebrectomies may be piecemeal or en bloc. As well, many reports of “en bloc spondylectomies” include violation of the underlying tumors. The literature supports the role of margin negative en bloc resections in maximizing the treatment outcome of primary spine tumors (12).
How the lack of ability to reliably obtain a surgical margin in spine tumors influences the differential prognosis of these patients is unclear but is hypothesized to account for at least of portion of the negative prognosis and lack of stratification seen with traditional staging protocols. For this reason, the AJCC 8th edition created new TNM categories for spine tumors which imply resectability and margin status. As there is a paucity of data regarding prognostic factors specific to primary spine tumors, these changes were made based largely on expert experience and opinion. Thus, they are best understood as an educated attempt to increase knowledge in this area. As data are accumulated in the National Cancer Data Bank and other registries, subsequent analysis will determine whether or not these new criteria have greater discriminate and predictive value.
8th edition staging system for spine tumors
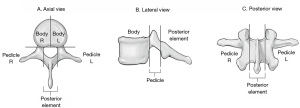
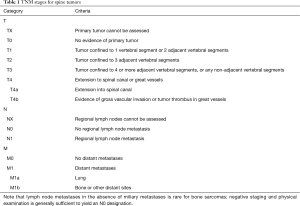
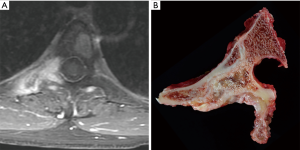
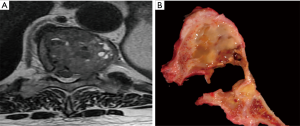
Figure 3 and Tables 1,2 present the new staging system for spine tumors. Note that the spine is divided into five segments; tumors in two segments are generally resectable with proper margins by experienced surgeons; tumors in three segments are likely resectable with a negative margin in many cases. More advanced tumors or those with significant epidural or retroperitoneal extension are challenging to resect with a true oncologic margin. A typical case for resection is shown in Figure 4, with tumor in two segments and no epidural extension. An experienced spine tumor surgeon can reliably resect a tumor of this nature. However, many cases will present as in Figure 5, with a wide margin of resection not possible because of epidural extension.
Full table
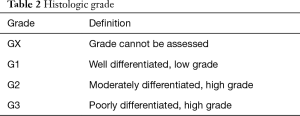
Full table
Note that the group formulating this new system considered the adoption of the WBB system (3). However, in an assessment by expert members of the Spine Oncology Study Group, this system had only modest interobserver agreement (18). Additionally, in practice, much of the staging classification in the National Cancer Databank is performed by pathologists rather than surgeons. Initial discussions among the working group formulating these new criteria anticipated worse reliability of this system with non-surgeons.
Traditionally, TNM categories were combined to provide a prognostic stage grouping for bone sarcomas. While this system continues for extremity sarcomas, in the 8th edition of the AJCC, there are no prognostic stage groupings for spine (or pelvic) tumors. This reflects the novel approach which is being taken to spine tumor staging with this new iteration of the AJCC. Further time and data collection will indicate which (if any) of these modifications improve discrimination of patient outcomes.
Limitations
Three primary limitations exist for this change in the AJCC staging criteria. First, little data exist to guide risk factors for poor outcome or limited resection ability in axial sarcomas and chordomas beyond expert opinion and experience (19). Thus, the criteria which were used to define the T categories used in this formulation are admittedly based primarily on experienced expert opinion rather than a firm evidence basis. The rare nature of these tumors and limited scientific literature prevent a more rigorous approach at this time.
Second, there remain tumors which will fall outside of the domains of these criteria. For example, locally advanced spinopelvic tumors which span multiple anatomic compartments remain poorly captured by the current criteria. Similarly, these criteria define tumor extension around a stereotypical thoracic or lumbar vertebra. While primary cervical bone tumors exist, they are rare. However, additional anatomic constraints regarding involvement of dominant or bilateral vertebral arteries will impact resectability of primary bone tumors of the cervical spine.
Finally, opportunities for non-surgical local control modalities continue to advance. In particular, the role of proton beam or other high precision radiotherapy techniques is an area of continued advance in the treatment of spine tumors (20). While the author’s personal experience with the use of proton beam therapy for definitive local control of spine sarcomas (other than Ewing’s) with adverse presentations has been poor, reports in the literature indicate that this may be an option for some patients in whom surgery is overly morbid or would be unreliable in obtaining a negative margin. Continued advancement and refinement in this field may mitigate the unfavorable surgical presentation of spine sarcomas and decrease the need for differential staging for these patients.
Conclusions
The recently adopted 8th edition staging manual of the AJCC redefine the staging of spine tumors in a distinct manner from extremity tumors. This change has been made in an attempt to identify prognostic factors for spine tumors separate from their analogous presentations in the extremity. Surgeons who treat patients with spine tumors should be aware of these changes for proper patient registration and classification.
Acknowledgments
None.
Footnote
Conflicts of Interest:The authors have no conflicts of interest to declare.
References
- Kniesl JS, Rosenberg AE, Anderson PM, et al. Bone. In: Amin MB, Edge S, Greene D, et al. editors. AJCC Cancer Staging Manual, 8th Edition. Springer: Chicago, 2018.
- Enneking WF, Spanier SS, Goodman MA. A system for the surgical staging of musculoskeletal sarcoma. Clin Orthop Relat Res 1980.106-20. [PubMed]
- Boriani S, Weistein JN, Biagini R. Primary bone tumors of the spine. Terminology and surgical staging. Spine 1997;22:1036-44. [Crossref] [PubMed]
- Fisher CG, Versteeg AL, Dea N, et al. Surgical management of spinal chondrosarcomas. Spine 2016;41:678-85. [Crossref] [PubMed]
- Ozaki T, Flege S, Liljenqvist U, et al. Osteosarcoma of the spine: experience of the cooperative osteosarcoma study group. Cancer 2002;94:1069-77. [Crossref] [PubMed]
- Schoenfeld AJ, Hornicek FJ, Pedlow FX, et al. Osteosarcoma of the spine: experience in 26 patients treated at the Massachusetts General Hospital. Spine J 2010;10:708-14. [Crossref] [PubMed]
- Schwab J, Gasbarrini A, Bandiera S, et al. Osteosarcoma of the mobile spine. Spine 2012;37:E381-6. [Crossref] [PubMed]
- Anderson ME. Update on survival in osteosarcoma. Orthop Clin North Am 2016;47:283-92. [Crossref] [PubMed]
- Pakos EE, Nearchou AD, Grimer RJ, et al. Prognostic factors and outcomes for osteosarcoma: an international collaboration. Eur J Cancer 2009;45:2367-75. [Crossref] [PubMed]
- Bacci G, Longhi A, Cesari M, et al. Influence of local recurrence on survival in patients with extremity osteosarcoma treated with neoadjuvant chemotherapy: the experience of a single institution with 44 patients. Cancer 2006;106:2701-6. [Crossref] [PubMed]
- Boriani S. Chondrosarcoma of the mobile spine: report on 22 cases. Spine 2000;25:804-12. [Crossref] [PubMed]
- Fisher CG, Saravanja DD, Dvorak MF, et al. Surgical management of primary bone tumors of the spine: validation of an approach the enhance cure and reduce local recurrence. Spine 2011;36:830-6. [Crossref] [PubMed]
- Fuchs B, Dickey ID, Yaszemski MJ, et al. Operative management of sacral chordoma. J Bone Joint Surg Am 2005;87:2211-6. [PubMed]
- Picci P, Sangiorgi L, Rougraff BT, et al. Relationship of chemotherapy-induced necrosis and surgical margins to local recurrence in osteosarcoma. J Clin Oncol 1994;12:2699-705. [Crossref] [PubMed]
- Pring ME, Weber KL, Unni KK, et al. Chondrosarcoma of the pelvis: a review of sixty-four cases. J Bone Joint Surg Am 2001;83:1630-42. [Crossref] [PubMed]
- Talac R, Yaszemski MJ, Currier BL, et al. Relationship between surgical margins and local recurrence in sarcomas of the spine. Clin Orthop Relat Res 2002.127-32. [Crossref] [PubMed]
- Murakami H, Tomita K, Kawahara N, et al. Complete segmental resection of the spine, including the spinal cord, for telangiectatic osteosarcoma. Spine 2006;31:E117-22. [Crossref] [PubMed]
- Chan P, Boriani S, Fourney DR, et al. An assessment of the reliability of the Enneking and Weinstein-Boriani-Biagini classifications for staging of primary spine tumors by the Spine Oncology Study Group. Spine 2009;34:384-91. [Crossref] [PubMed]
- Yamazaki T, McLoughlin GS, Patel S, et al. Feasibility and safety of en bloc resection for primary spine tumors: a systematic review by the Spine Oncology Study Group. Spine 2009;34:S31-8. [Crossref] [PubMed]
- DeLaney TF, Liebsch NJ, Pedlow FX, et al. Long-term results of phase II study of high dose photon/proton radiotherapy in the management of spine chordomas, chondrosarcomas, and other sarcomas. J Surg Oncol 2014;110:115-22. [Crossref] [PubMed]


