
Comparison of posterior corneal elevation after SMILE and FS-LASIK in correcting myopia over −9.0 diopters
Introduction
Safety and stability have always been a major concern in evaluating corneal refractive surgery, especially in patients with high myopia. Researchers have demonstrated that iatrogenic keratectasis is prone to occur in patients with high myopia, as they require deeper ablation, and that the initial sign of keratectasis is protrusion of the posterior corneal surface (1). According to published studies, investigating posterior corneal elevation has proven to be an effective way to evaluate posterior corneal stability (2).
Corneal surgical correction for high myopia provides distinctive advantages over phakic intraocular lens (IOL) implantation; for instance, simple postoperative administration and low incidence of complications associated with intraocular surgery (3). Lindbohm evaluated a 2- to 5-year result of patients who underwent laser in situ keratomileusis (LASIK) with at least -9 diopters (D) preoperatively (4). The author proposed that LASIK is a safe alternative for the treatment of super high myopia with safety precautions and careful patient selection. Additionally, Alió conducted a longer follow-up (10-year) study of LASIK for correction of myopia up to −10 D, and demonstrated that LASIK was an effective and predictable procedure resulting in slight regression (5).
Recently, femtosecond lasers have been widely applied in the field of ophthalmology, especially in refractive surgery. It was initially used for the creation of corneal flaps in LASIK, followed by corneal stroma ablation by an excimer laser. The technology combining the excimer laser and femtosecond laser is known as femtosecond laser-assisted laser in situ keratomileusis (FS-LASIK) (6). Small incision lenticule extraction (SMILE), solely utilizing the femtosecond laser, is a new type of corneal refractive surgery free of the creation of a flap (7). Reinstein performed a retrospective study of high myopic FS-LASIK eyes between −8.00 and −14.25 D, and demonstrated that FS-LASIK could achieve high efficacy and safety for these patients (8). Moreover, in our previous study, SMILE was shown to be as safe and effective as FS-LASIK in the treatment of myopia over −10.00 D (9).
The aim of the current study was to evaluate posterior corneal stability both after SMILE and FS-LASIK at a 6-month follow-up, and to compare the two different procedures.
We present the following article in accordance with the STROBE reporting checklist (available at http://dx.doi.org/10.21037/atm-20-5165).
Methods
Patients
The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by the Ethics Committee of Fudan University Eye and ENT Hospital Review Board (Shanghai, China, No. KJ2008-10), and informed consent was obtained from all the patients.
In this prospective comparative study, 82 eyes of 82 patients who were scheduled for refractive correction from June 2015 to June 2016 at the Department of Ophthalmology, Eye and ENT Hospital of Fudan University (Shanghai, China) were recruited. Eyes were randomly assigned to the SMILE group (45 eyes) or FS-LASIK group (37 eyes). The patients had no ocular disease other than refractive error and met the inclusion criteria. Subjects participating in the study were 18 to 45 years of age with a preoperative manifest spherical equivalent over –9.00 D.
The mean preoperative manifest spherical equivalent was −10.43±0.92 D (range: −9.00 to −13.00 D) in the SMILE group and −10.97±1.37 D (range: −9.00 to −13.25 D) in the FS-LASIK group. All patients underwent a comprehensive preoperative ophthalmologic examination, including Pentacam HR imaging, slit-lamp examination, measurement of uncorrected distance visual acuity (UDVA), corrected distance visual acuity (CDVA), intraocular pressure, and other examinations. The detailed data are shown in Table 1.
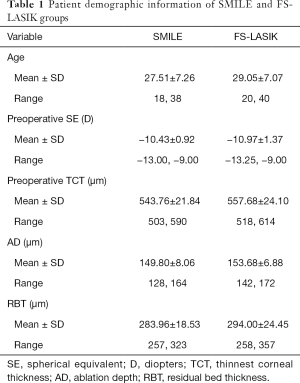
Full table
Surgical procedure
Small incision lenticule extraction
The VisuMax femtosecond laser system (Carl Zeiss Meditec AG, Germany) was used to perform all of the surgeries. After topical anesthesia, the patient was positioned under the curved contact glass and instructed to focus on the internal target light. The surgeon then achieved correct corneal centration and initiated suction, followed by femtosecond laser scanning. Once the laser scanning was completed, the surgeon inserted a spatula into the cornea, dissecting the lenticule interface, and manually extracted the lenticule. The femtosecond laser settings were as follows: repetition rate of 500 kHz, 110 µm intended cap thickness, 5.8 to 6.5 mm optical zone (lenticule diameter), 7.3 to 7.5 mm cap diameter, and a 2-mm side cut at the 12 o’clock point. The same experienced surgeon performed all procedures (XZ).
Femotosecond-assisted laser in situ keratomileusis
The same experienced surgeon performed all of the procedures. All flaps, created using the same VisuMax femtosecond laser system, had a superior hinge. After the flap was scanned, a spatula was inserted to lift it. Stromal ablation was performed with a Mel-80 excimer laser system (Carl Zeiss Meditec AG, Germany). The flap diameter was set at 7.5 mm, and 110 µm for the flap thickness. The optical zone varied between 5.75 and 6.25 mm according to the refractive errors and corneal thickness. A bandage contact lens was applied to protect the eye, which was removed the next day.
Pentacam Scheimpflug imaging
All eyes were examined using the Pentacam imaging system (Oculus GmbH, Wetzlar, Germany). The patient was instructed to position their head on the headrest and fixate the target light. After attaining alignment, the device captured 25 images and recorded 12,500 elevation points automatically within 2 seconds. To avoid miscalculations of poor imaging, the quality of each measurement was shown in the specification window, and only results with “OK” statements were accepted. The examination was duplicated if the statement did not meet the requirement (marked yellow or red). Only maps with at least 10 mm of corneal coverage and no deduced data in the central 9-mm zone were accepted.
Postoperative examination
Follow-up appointments were scheduled at 1 day, 1 month, and 6 months after surgery (eyes undergoing FS-LASIK were followed at 1 day and 6 months postoperatively). Postoperative examinations included Pentacam imaging examinations, slit-lamp examination, measurement of UDVA, CDVA, spherical equivalent refraction, and intraocular pressure.
Data collection
Elevation data of the posterior corneal surfaces were obtained using Pentacam software. The reference best-fit sphere (BFS) was defined in the center 8.0-mm region of the cornea and determined by the preoperative data, so it was the same across all images. For points above the reference, values were positive; for points below the reference, values were negative. Calculated values were obtained from 27 points in the central 6-mm zone as follows: 1 point at the center, 4 points at 1 mm distance from the center along 45°, 135°, 225°, and 315° meridians (0°, defined as the horizontal semi-meridian on the right, and rotating counterclockwise in both eyes), 8 points at 2 mm distance from the center at 0°, 45°, 90°, 135°, 180°, 225°, 270°, 315°, and the other 14 points at 3 mm distance from the center along 15°, 45°, 75°, 90°, 105°, 135°, 165°, 195°, 225°, 255°, 270°, 285°, 315°, and 345°. Posterior corneal elevation in the central 4mm area and in various optical zones (2-, 4-, and 6-mm diameter) was calculated as the mean value from determined points in the corresponding area. Changes in the posterior elevation were found by subtracting preoperative data from postoperative data (difference elevation map). The change in elevation was the shift of the posterior corneal surface, with a positive number indicating ectatic change. Elevation data were recorded in an Excel Spreadsheet (Microsoft Corp, Redmond, WA, USA) for further analysis.
Statistical analysis
The descriptive results contained the mean and the standard deviation. The Kolmogorov-Smirnov normality test and test for homogeneity of variances were performed for all data. The analysis of variance (ANOVA) for repeated measures with the Bonferroni correction was employed to compare pre- and postoperative values. If the data were not suitable for ANOVA analysis, we used the Friedman’s rank test for k correlated samples. Bivariate normal analysis was performed before the correlation test. The Pearson or Spearman correlation test was applied subsequently to determine the association between the change in posterior corneal surface 6 months after surgery and residual bed thickness (RBT). Statistical analyses were performed using SPSS ver. 20.0 (SPSS Inc., Chicago, IL, USA). A P value <0.05 was considered a statistically significant difference.
Patient and public involvement
The role of the patients in this study was participants. They were not involved in the development of the research question and outcome measures, the recruitment of subjects, and the conduct of the study. They were not involved in the design of this study. The results of the study will be disseminated to all participants by email. No specific patient advisers were involved in the design or conduct of the study.
Results
Visual outcomes
All surgeries were completed successfully. The safety index and efficacy index were 1.20 and 1.00, respectively, in the SMILE group, and 1.10 and 0.90, respectively, in the FS-LASIK group. At the last follow-up, 80% (36/45) of eyes in the SMILE group and 65% (24/37) in the FS-LASIK group were within ±1.00 D; a best corrected distance visual acuity (BDVA) of 0.8 or better was achieved in 100% of eyes.
Small incision lenticule extraction
The posterior corneal elevation of all points in various annulus of the cornea is shown in Figure 1. As seen in Table 2, the mean values of central posterior corneal elevation at different follow-ups were similar to each other, and no notable difference was observed between the times. The same results were also found for the 2-mm areas, as no statistical difference existed among different times. A significant statistical difference was revealed between pre- and postoperative 1 day in the 6-mm area. No significant difference existed among measurements at different times for the central 4-mm optical zone.
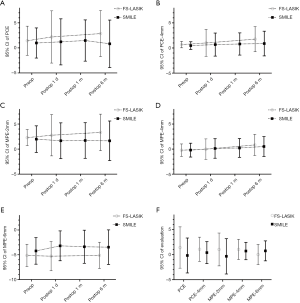
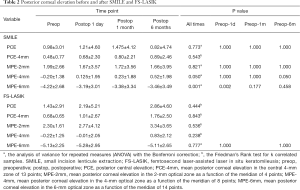
Full table
Femotosecond-assisted laser in situ keratomileusis
Changes in posterior corneal elevation in eyes that underwent FS-LASIK had some differences when compared to those who underwent SMILE. The posterior corneal elevation at the central point, various corneal rings, and the central 4-mm area showed a slight forward displacement but no statistical significance (Figure 1, Table 2). Changes in posterior corneal elevation after FS-LASIK were greater than after SMILE, with no statistical significance (P≥0.07) (Table 3).

Full table
Correlations
In the FS-LASIK group, RBT was significantly positively correlated with changes in posterior corneal elevation in the 3-mm radius of the corneal annulus. For eyes that underwent SMILE, RBT was positively correlated with changes in the 3-mm radius corneal ring. As for the central corneal area in the SMILE group, including the posterior corneal elevation in the center, the 1-mm radius corneal circle, and the central 4-mm optical zone, RBT was significantly negatively related to changes in the values (Figure 2, Table 3).
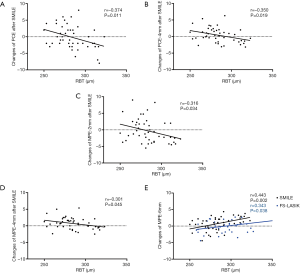
Discussion
It has been theoretically proven that the greater the myopia is, the thicker the corneal ablation required, which may result in less corneal thickness with loss of corneal biomechanical stability leading to corneal protrusion after the procedure (10). Therefore, we studied the changes in the posterior corneal surface after FS-LASIK and SMILE in super high myopia.
First, no statistical significant difference was noted between the 6-month postoperative and baseline values when analyzing values in various zones. The results indicated that the posterior corneal surface remained stable after FS-LASIK and SMILE with no corneal forward displacement in super high myopia. In our previous study, we analyzed the changes in posterior corneal elevation in high myopia. The results revealed that the posterior corneal surface remained stable after SMILE (11). Considering patients who underwent FS-LASIK, Grewal evaluated the posterior corneal stability in the central 5-mm zone postoperatively and demonstrated no significant changes following the procedure (12). Cagini also observed that the posterior corneal surface did not have a forward displacement after FS-LASIK during follow-up (13). The results in the current study are in good agreement with those of the above studies. However, changes of posterior corneal elevation in longer periods for correction of myopia over −9.0 D is still unknown and worthwhile investigated.
Although no statistical significance existed when comparing the changes in posterior corneal elevation after 6 months between the two groups, there were still some differences that should not be ignored. The mean changes in posterior corneal elevation in various areas after SMILE were less pronounced within 1 µm; however, greater changes after FS-LASIK were shown as the values were around 1 µm in almost all regions. A previous study also found that the change in posterior corneal elevation after FS-LASIK was greater than that after SMILE (14). The discrepancy in the wound healing response and biomechanical properties postoperatively may account for this finding. Dong has conducted a comparative study to evaluate corneal wound healing and inflammatory course after these two surgeries, and found that SMILE resulted in less keratocyte apoptosis, proliferation, and inflammation compared with FS-LASIK (15). SMILE may result in a lower wound healing response, which can lead to more stable posterior corneal elevation due to the following reasons. On one hand, compared with the excimer laser, which uses ultraviolet light to break molecular bonds in the corneal stroma, the femtosecond laser produces ultrashort light pulses of high frequencies with a lower energy level, permitting photo-disruption of the corneal tissue at a precise depth (16). On the other hand, SMILE possesses distinct advantages over FS-LASIK for a 2-mm small incision and is free of lifting the cap, leaving most of the anterior stromal lamellae intact postoperatively (17). Reinstein developed a mathematical model to calculate the total stromal tensile strength following photorefractive keratectomy (PRK), LASIK, and SMILE, and concluded that SMILE possesses the greatest tensile strength (18). Based on another comparison experiment, Sinha Roy inferred that SMILE could theoretically afford less biomechanical risk than LASIK (19).
In the SMILE group, changes in the posterior corneal elevation in the central area were found to be moderately negatively correlated with RBT, whereas changes in the peripheral area were positively correlated with RBT. Similar results in the peripheral zone were also noted in the FS-LASIK group. The correlation between changes in posterior corneal elevation and RBT remains controversial. Grewal et al. revealed no correlation between change in posterior corneal elevation and RBT in either of three corneal refractive surgery groups (FS-LASIK, LASIK, and LASEK) (12). Sun also demonstrated that the RBT had no correlation with the amount of forward displacement (20). In contrast, another study reported that eyes with a thinner RBT were more susceptible to show a greater positive change in posterior corneal elevation in the central area after SMILE, which is consistent with our results (14). It is rational to infer that several factors may lead to different results: with a longer follow-up period, smaller sample size, and larger analyzed zone (especially for data covering both the central and peripheral areas at the same count), the correlation was weaker or even not statistically significant.
There are limitations to our study: the sample size was small and the observation time was short. Future studies will include a lager sample size and a longer follow-up, which will provide more information on the posterior corneal surface changes after these two surgeries.
In the current study, our results demonstrated that both SMILE and FS-LASIK present a promising advance in the correction of myopic over −9.0 D. At the final follow-up visit of 6 months, all recruits in the two groups exhibited a stable posterior corneal surface.
Acknowledgments
We would like to thank Editage (www.editage.cn) for English language editing.
Funding: This work was supported by Shanghai Sailing Program (Grant No. 20YF1405200 to YZ), the National Natural Science Foundation of China (Grant No. 82000932 to YZ), the National Natural Science Foundation of China (Grant No. 81770955 to XTZ), Project of Shanghai Science and Technology (Grant No. 20410710100 to XTZ) and Joint Research Project of New Frontier Technology in Municipal Hospitals (Grant No. SHDC12018103 to XTZ). The funding organization had no role in the design or conduct of this research.
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at http://dx.doi.org/10.21037/atm-20-5165
Data Sharing Statement: Available at http://dx.doi.org/10.21037/atm-20-5165
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/atm-20-5165). The authors have no conflicts of interest to declare.
Ethics Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by the Ethics Committee of Fudan University Eye and ENT Hospital Review Board (Shanghai, China, No. KJ2008-10) and informed consent was taken from all the patients.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Pallikaris IG, Kymionis GD, Astyrakakis NI. Corneal ectasia induced by laser in situ keratomileusis. J Cataract Refract Surg 2001;27:1796-802. [Crossref] [PubMed]
- Nishimura R, Negishi K, Saiki M, et al. No forward shifting of posterior corneal surface in eyes undergoing LASIK. Ophthalmology 2007;114:1104-10. [Crossref] [PubMed]
- Stonecipher KG, Kezirian GM, Stonecipher M. LASIK for -6.00 to -12.00 D of myopia with up to 3.00 D of cylinder using the ALLEGRETTO WAVE: 3- and 6-month results with the 200- and 400-Hz platforms. J Refract Surg 2010;26:S814-8. [Crossref] [PubMed]
- Lindbohm N, Tuisku IS, Tervo TM. LASIK for myopia of -9.00 to -17.00 D with the VISX STAR S2: 2- to 5-year follow-up. J Refract Surg 2009;25:195-200. [Crossref] [PubMed]
- Alio JL, Muftuoglu O, Ortiz D, et al. Ten-year follow-up of laser in situ keratomileusis for myopia of up to -10 diopters. Am J Ophthalmol 2008;145:46-54. [Crossref] [PubMed]
- Holzer MP, Rabsilber TM, Auffarth GU. Femtosecond laser-assisted corneal flap cuts: morphology, accuracy, and histopathology. Invest Ophthalmol Vis Sci 2006;47:2828-31. [Crossref] [PubMed]
- Sekundo W, Kunert KS, Blum M. Small incision corneal refractive surgery using the small incision lenticule extraction (SMILE) procedure for the correction of myopia and myopic astigmatism: results of a 6 month prospective study. Br J Ophthalmol 2011;95:335-9. [Crossref] [PubMed]
- Reinstein DZ, Carp GI, Archer TJ, et al. Long-term Visual and Refractive Outcomes After LASIK for High Myopia and Astigmatism From -8.00 to -14.25 D. J Refract Surg 2016;32:290-7. [Crossref] [PubMed]
- Qin B, Li M, Chen X, et al. Early visual outcomes and optical quality after femtosecond laser small-incision lenticule extraction for myopia and myopic astigmatism correction of over -10 dioptres. Acta Ophthalmol 2018;96:e341-6. [Crossref] [PubMed]
- Baek T, Lee K, Kagaya F, et al. Factors affecting the forward shift of posterior corneal surface after laser in situ keratomileusis. Ophthalmology 2001;108:317-20. [Crossref] [PubMed]
- Zhao Y, Li M, Zhao J, et al. Posterior Corneal Elevation after Small Incision Lenticule Extraction for Moderate and High Myopia. PLoS One 2016;11:e0148370. [Crossref] [PubMed]
- Grewal DS, Brar GS, Grewal SP. Posterior corneal elevation after LASIK with three flap techniques as measured by Pentacam. J Refract Surg 2011;27:261-8. [Crossref] [PubMed]
- Cagini C, Messina M, Lupidi M, et al. Posterior Corneal Surface Stability after Femtosecond Laser-Assisted Keratomileusis. J Ophthalmol 2015;2015:184850. [Crossref] [PubMed]
- Wang B, Zhang Z, Naidu RK, et al. Comparison of the change in posterior corneal elevation and corneal biomechanical parameters after small incision lenticule extraction and femtosecond laser-assisted LASIK for high myopia correction. Cont Lens Anterior Eye 2016;39:191-6. [Crossref] [PubMed]
- Dong Z, Zhou X, Wu J, et al. Small incision lenticule extraction (SMILE) and femtosecond laser LASIK: comparison of corneal wound healing and inflammation. Br J Ophthalmol 2014;98:263-9. [Crossref] [PubMed]
- Sarayba MA, Ignacio TS, Binder PS, et al. Comparative study of stromal bed quality by using mechanical, IntraLase femtosecond laser 15- and 30-kHz microkeratomes. Cornea 2007;26:446-51. [Crossref] [PubMed]
- Moshirfar M, McCaughey MV, Reinstein DZ, et al. Small-incision lenticule extraction. J Cataract Refract Surg 2015;41:652-65. [Crossref] [PubMed]
- Reinstein DZ, Archer TJ, Randleman JB. Mathematical model to compare the relative tensile strength of the cornea after PRK, LASIK, and small incision lenticule extraction. J Refract Surg 2013;29:454-60. [Crossref] [PubMed]
- Sinha Roy A, Dupps WJ Jr, Roberts CJ. Comparison of biomechanical effects of small-incision lenticule extraction and laser in situ keratomileusis: finite-element analysis. J Cataract Refract Surg 2014;40:971-80. [Crossref] [PubMed]
- Sun HJ, Park JW, Kim SW. Stability of the posterior corneal surface after laser surface ablation for myopia. Cornea 2009;28:1019-22. [Crossref] [PubMed]


