
Coronavirus disease 19 positivity after negative results from six consecutive tests
On March 11, 2020, the World Health Organization declared the novel coronavirus (COVID-19) outbreak a global pandemic, and it has spread to more than 200 countries. Approximately 2–5% of COVID-19 cases result in death due to massive alveolar damage and progressive respiratory failure (1). Because the 2019 novel coronavirus (2019-nCoV) is highly infectious, patient isolation is important for preventing secondary infections (1). For management of COVID-19, accurate diagnosis is essential. Real-time polymerase chain reaction (RT-PCR) is recommended as the final confirmatory test by the World Health Organization, as it helps identify the presence of the virus itself. Currently, RT-PCR is the standard diagnostic technique used by countries worldwide (2). However, clinicians in many hospitals have reported false-negative RT-PCR results (3). Here, we raise awareness on false-negative PCR results for 2019-nCoV by reporting a related case.
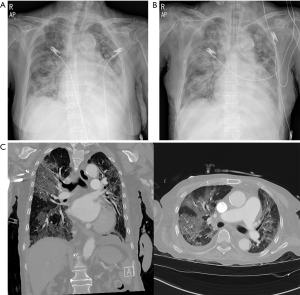
An 86-year-old woman, with a history of chronic kidney disease, diabetes, hypertension, and major depressive disorder visited the emergency room (ER) of our university hospital complaining of general weakness for 2 days. Upon arrival at the ER, her O2 saturation was 70%. Her other vital signs were as follow: blood pressure, 100/50 mmHg; heart rate, 70 beats/min; body temperature, 35.4 °C; and respiratory rate, 20 breaths/min. Her initial laboratory test results from the ER were as follows: white blood cell count, 2.75k/µL (differential neutrophil and lymphocyte counts, 83.4% and 11.0%, respectively), and C-reactive protein level, 20.027 mg/L. Initial chest X-ray (CXR) showed severe haziness in both lung fields (Figure 1A). A pharyngeal swab was performed, and RT-PCR (Allplex™ 2019-nCoV Assay, Seegene®, South Korea) for 2019-nCoV yielded negative results. The patient was intubated and received ventilator support. On the next day, CXR revealed aggravated haziness (Figure 1B), and chest computed tomography (CT) showed multifocal ground glass opacities with a crazy paving appearance in both lung fields (Figure 1C). RT-PCR for 2019-nCoV was repeated and yielded negative results again. Treatment with 10 g of Piperacillin/Tazobactam intravenous daily was started empirically. On hospital day 3, a third RT-PCR for 2019-nCoV was carried out and yielded negative results. Because the diagnosis of COVID-19 was not confirmed, the patient was admitted to the non-isolated intensive care unit (ICU) for further management. Once transferred on hospital day 3, a fourth RT-PCR for 2019-nCoV was carried out and yielded negative results. Although the RT-PCR tests repetitively yielded negative results, COVID-19 could not be completely ruled out because COVID-19 was prevalent and CT findings suggested viral pneumonia. Therefore, the patient was started on 800 mg of Lopinavir/Ritonavir (Kaletra) and 200 mg of Hydroxychloroquine sulfate (Oxiklorin) per oral daily from hospital day 3. On hospital day 4, the fifth and sixth RT-PCR tests were performed, both yielding negative results. However, on hospital day 5, a seventh RT-PCR test for 2019-nCoV yielded positive results. Consecutively, an eighth RT-PCR test on hospital day 5 also yielded positive results. Eventually, the patient was diagnosed as having COVID-19 and moved to an isolated ICU. Considering the fatality rate of COVID-19, specifically among patients with underlying disease, the other patients in the non-isolated ICU had been exposed and put at risk for developing COVID-19. Fortunately, during the following two weeks, any patients admitted to the non-isolated ICU and any workers in the non-isolated ICU admitted in non-isolated ICU were not diagnosed as a COVID-19. On hospital day 13, the patient recovered and was moved to an isolated general ward. She continued receiving Hydroxychloroquine sulfate (Kaletra was stopped after 8 days) treatment.
The diagnosis of COVID-19 is confirmed by RT-PCR (2-4). Although the sensitivity of RT-PCR has not been clearly determined, it is known to be around 70–80% (3,4). Therefore, the false negative rate per RT-PCR test may range from 20 to 30%. Additionally, most RT-PCR kits use only 2–3 primers for 2019-nCoV gene detection. Considering that 2019-nCoV is prone to mutation, diagnosis relying on only a few primers may be one of the reasons for the false-negative results.
Importantly, negative RT-PCR results may lead to COVID-19 patients not being isolated. Therefore, transmission of 2019-nCoV to other people can occur, potentially causing mass infection. Considering this, clinicians should be aware of the possibility of false-negative PCR results for 2019-nCoV. Chest CT is reported to have a high sensitivity for COVID-19 diagnosis (3). Therefore, clinicians should make a diagnosis of COVID-19 on the basis of chest CT findings and clinical features as well as RT-PCR results. When RT-PCR results are negative but image findings and symptoms are indicative of COVID-19, we recommend isolating patients from other people and performing repeat RT-PCR tests with close observation of the patients’ symptoms.
Acknowledgments
Funding: This work was supported by the 2020 Yeungnam University Research Grant.
Footnote
Provenance and Peer Review: This article was a free submission to the journal. The article did not undergo external peer review.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/atm-20-2994). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Written informed consent was obtained from the patient for publication of this study and any accompanying images.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Kucharski AJ, Russell TW, Diamond C, et al. Early dynamics of transmission and control of COVID-19: a mathematical modelling study. Lancet Infect Dis 2020;20:553-8. [Crossref] [PubMed]
- Li D, Wang D, Dong J, et al. False-Negative Results of Real-Time Reverse-Transcriptase Polymerase Chain Reaction for Severe Acute Respiratory Syndrome Coronavirus 2: Role of Deep-Learning-Based CT Diagnosis and Insights from Two Cases. Korean J Radiol 2020;21:505-8. [Crossref] [PubMed]
- Ai T, Yang Z, Hou H, et al. Correlation of Chest CT and RT-PCR Testing for Coronavirus Disease 2019 (COVID-19) in China: A Report of 1014 Cases. Radiology 2020;296:E32-40. [Crossref] [PubMed]
- Fang Y, Zhang H, Xie J, et al. Sensitivity of Chest CT for COVID-19: Comparison to RT-PCR. Radiology 2020;296:E115-7. [Crossref] [PubMed]


