Nosocomial infections among patients with COVID-19, SARS and MERS: a rapid review and meta-analysis
Introduction
COVID-19 is a respiratory infectious disease caused by a novel coronavirus, SARS-CoV-2. The first batch of COVID-19 patients were found in December 2019 (1). The disease is mainly transmitted through respiratory droplets and close contact, and all people are susceptible to it (2). SARS-CoV-2 is highly contagious (3), and has quickly spread to most countries and regions of the world. COVID-19 has become a global pandemic and has received great attention from all over the world (4,5). As of April 7, 2020, 1,214,466 confirmed cases of COVID-19 have been found in 211 countries and regions, causing 67,767 deaths (6).
The main clinical manifestations of COVID-19 are cough, fever and complications such as acute respiratory distress syndrome (1). Disease clusters and nosocomial infections have been reported (7,8). The proportion of nosocomial infections is high among diagnosed infections, and medical staff are at high risk of infection (8). One study on 44,672 patients showed that health workers accounted for 3.8% of the COVID-19 cases and five health workers died as a result of the infection (9). There is still no specific medicine for COVID-19, so preventing nosocomial infections is crucial.
This study compares the incidence of nosocomial infections during the COVID-19, SARS and MERS epidemics and analyzes the characteristics of the nosocomial infection, to enhance the understanding of nosocomial infection among medical and non-medical staff. We present the following article in accordance with the PRISMA reporting checklist (available at http://dx.doi.org/10.21037/atm-20-3324).
Methods
Search strategy
An experienced librarian searched the following databases from their inception to March 31, 2020 in the following electronic databases (10): the Cochrane Library, MEDLINE (via PubMed), EMBASE, Web of Science, CBM (China Biology Medicine disc), CNKI (China National Knowledge Infrastructure), and Wanfang database. We made no restrictions on language or publication status. We used the following search formula is as follow: (“Novel coronavirus” OR “2019-novel coronavirus” OR “Novel CoV” OR “2019-nCoV” OR “COVID-19” OR “SARS-CoV-2” OR “Middle East Respiratory Syndrome” OR “MERS” OR “MERS-CoV” OR “Severe Acute Respiratory Syndrome” OR “SARS” OR “SARS-CoV” OR “SARS-Related” OR “SARS-Associated”) AND (“Cross Infection” OR “Cross Infections” OR “Healthcare Associated Infections” OR “Healthcare Associated Infection” OR “Health Care Associated Infection “ OR “Health Care Associated Infections” OR “Hospital Infection” OR “Nosocomial Infection” OR “Nosocomial Infections” OR “Hospital Infections” OR “hospital-related infection” OR “hospital-acquired infection”). We also searched clinical trial registry platforms [the World Health Organization Clinical Trials Registry Platform (http://www.who.int/ictrp/en/), US National Institutes of Health Trials Register (https://clinicaltrials.gov/)], Google Scholar (https://scholar.google.nl/), preprint platform [medRxiv (https://www.medrxiv.org/), bioRxiv (https://www.biorxiv.org/) and SSRN (https://www.ssrn.com/index.cfm/en/)] and reference lists of the included reviews to find unpublished or further potential studies. Finally, we contacted experts in the field to identify relevant trials. The search strategy was also reviewed by another information specialist. The details of the search strategy can be found in the Supplement I.
Inclusion and exclusion criteria
We included case series studies and case reports about the proportion of cases of COVID-19, SARS and MERS who were infected in health facilities, about infections among medical staff and outbreaks in hospitals. Abstract, letter, new, guideline, articles for which we could not access all relevant data or full text were excluded.
Study selection
After eliminating duplicates, two reviewers (Y Gao and X Wang) independently selected the relevant studies in two steps with the help of the EndNote software. Discrepancies were settled by discussion or consulting a third reviewer (Q Zhou). In the first step, all titles and abstracts were screened using pre-defined criteria. In the second step, full-texts of the potentially eligible and unclear studies were reviewed to decide about final inclusion. All reasons for exclusion of ineligible studies were recorded. The process of study selection was documented using a PRISMA flow diagram (11).
Data extraction
Two reviewers (R Liu and X Wang) extracted the data independently using a standardized data collection table. Any differences were resolved by consensus, and a third auditor checked the consistency and accuracy of the data. The following data were extracted: (I) basic information: title, first author, country, year of publication, and type of study; (II) population baseline characteristics: age and sex distribution, and sample size; and (III) the proportion of nosocomial infections, the proportion of patients with occupation of medical staff, and for studies on hospital outbreaks, the number of index cases and total infections.
Risk of bias assessment
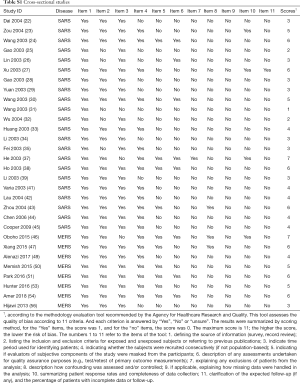
Two researchers (Z Wang and Q Shi) independently assessed the potential bias in each included study. The included studies were evaluated using appropriate assessment scales depending on the study type: for case control studies, the Newcastle-Ottawa Scale (NOS) (12), for cross-sectional studies and epidemiological surveys, the methodology evaluation tool recommended by the Agency for Healthcare Research and Quality (AHRQ) (13), and for case reports and case series, we used a methodology evaluation tool recommended by National Institute for Health and Care Excellence (NICE) (14).
Data synthesis
We performed a meta-analysis of proportions for dichotomous outcomes (nosocomial infection among the confirmed cases, and infections among the health care workers), reporting the effect size (ES) with 95% confidence intervals (CI) by using random-effects models. Two-sided P values <0.05 were considered statistically significant. Heterogeneity was defined as P<0.10 and I2>50%. All analyses were performed in STATA version 14. All results are limited to 0–100%.
Quality of the evidence assessment
Two reviewers (Z Wang and Q Shi) assessed the quality of evidence independently using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) (15,16). We produced a “Summary of Findings” table using the GRADEpro software. This table includes overall grading of evidence body for each prespecified outcome that is accounted in a meta-analysis. The overall quality can be downgraded for five considerations (study limitations, consistency of effect, imprecision, indirectness, and publication bias) and upgraded for three considerations (large magnitude of effect, dose-response relation and plausible confounders or biases). The overall quality of evidence will be classified as high, moderate, low or very low, which reflecting to what extent that we can be confident the effect estimates are correct.
As COVID-19 is a public health emergency of international concern and the situation is evolving rapidly, our study was not registered in order to speed up the process (17).
Results
Characteristics and quality of included studies
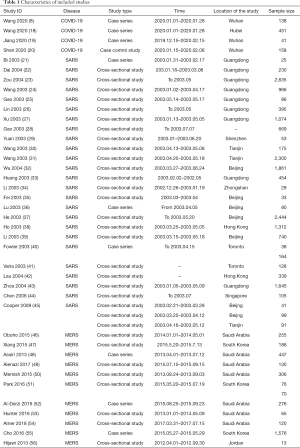
Our initial search revealed 2,626 articles, of which 2,598 were left after deleting the duplicates (Figure 1). After review the titles and abstracts, we screened the full texts of 66 articles, of which 40 were finally included (Table 1). Four studies were about COVID-19 (8,18-20), 25 studies about SARS (21-45), and 11 studies about MERS (46-56) (Table 1). Sixteen studies described the number of nosocomial infections in a selected patient population, 16 studies described the situation of nosocomial infections among the staff of medical institutions, and 13 studies reported the number of nosocomial infections caused by one or more than one patient. The quality of included studies was very poor: all cross-sectional studies scored less than 8 out of 11 in the evaluation by the AHRQ tool, half case series studies scored less than 5 out of 8 in the evaluation by the NICE tool, and only one case-control study scored 6 by the NOS tool. The details of the risk of bias of included studies can be found in the Supplement II (Tables S1-S3).
Full table
Full table
Full table

Full table
Nosocomial infections among confirm cases
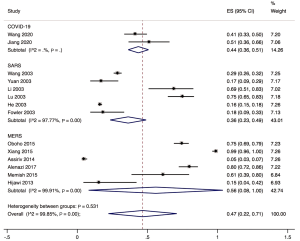
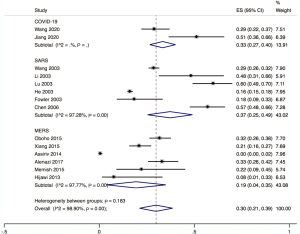
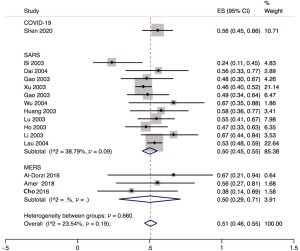
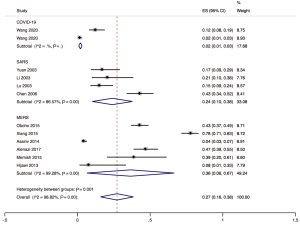
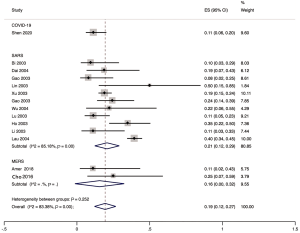
The proportion of nosocomial infections was 44.0% (95% CI: 0.36 to 0.51; I2=0.00%) among COVID-19 patients in the early outbreak, 36.0% (95% CI: 0.23 to 0.49; I2=97.8%) among SARS patients, and 56.0% (95% CI: 0.08 to 1.00; I2=99.9%) among MERS patients (Figure 2). Thirty-three percent (95% CI: 0.27 to 0.40; I2=0.00%) of patients with COVID-19 were medical staff, and 2.0% (95% CI: 0.01 to 0.03; I2=0.00%), were nosocomial infections among people other than medical staff (such as inpatients or visitors). The corresponding proportions among SARS patients were 37.0% (95% CI: 0.25 to 0.49; I2=97.3%) and 24.0% (95% CI: 0.10 to 0.38; I2=86.6%), and 19.0% (95% CI: 0.04 to 0.35; I2=97.8%) and 36.0% (95% CI: 0.06 to 0.67; I2=99.3%) among MERS patients (Figures 3,4).
Infection among the health care workers
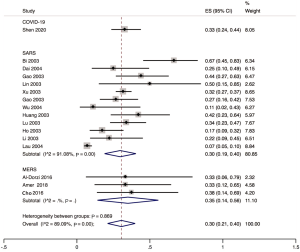
Twenty studies mentioned infection among the health workers, of which sixteen studies described the occupational composition of infected health care workers. Doctors accounted for 33.0% (95% CI: 0.24 to 0.44), nurses 56.0% (95% CI: 0.45 to 0.66), and other staff (such as carers, cleaners, hospital support staff) 11.0% (95% CI: 0.06 to 0.20) of COVID-19 cases among hospital staff in the early outbreak in Wuhan. For SARS, 30.0% (95% CI: 0.19 to 0.40; I2=91.1%) of the cases among hospital workers were doctors, 50.0% (95% CI: 0.45 to 0.55; I2=38.8%) nurses, and 21.0% (95% CI: 0.12 to 0.29; I2=85.2%) others. For MERS, for the corresponding proportions were 35.0% (95% CI: 0.14 to 0.56; I2=0.00%), 50.0% (95% CI: 0.29 to 0.71; I2=0.00%) and 16.0% (95% CI: 0.00 to 0.32; I2=0.00%). For all three conditions combined, the proportion of doctors among infected hospital staff was 30.0%, 51.0% for the proportion of nurses, and 19.0% for the proportion of others (Figures 5-7).
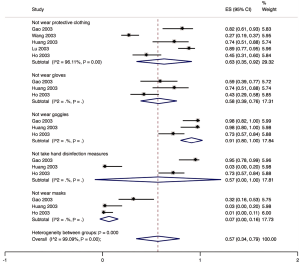
Five studies described the protective measures of medical staff infected with SARS in hospital. Sixty-three percent (95% CI: 0.35 to 0.92; I2=96.1%) of the infected staff did not wear protective clothing), 58.0% (95% CI: 0.39 to 0.76; I2=0.00%) did not use gloves, 91.0% (95% CI: 0.80 to 1.00; I2=0.00%) did not wear goggles; 57.0% (95% CI: 0.00 to 1.00; I2=0.00%) did not take any hand disinfection measures), and 7.0% (95% CI: 0.00 to 0.16; I2=0.00%) did not wear masks (Figure 8). One study described that among the 22 infected medical workers, 21 had no shoe cover. One study described that of 53 infected health workers, 47 wore cloth masks.
Outbreaks in the hospitals
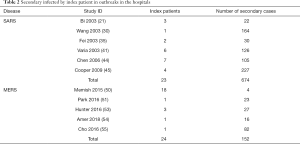
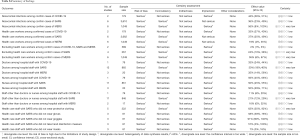
Six studies described SARS outbreaks, and five studies MERS outbreaks that happened in hospitals. The SARS studies reported on 23 patients, causing a total of 674 infections in hospitals, with an average of 29.3 infections per index patient. The MERS studies reported 24 patients causing 152 infections in hospitals, with an average of 6.3 infections per index patient (Table 2).
Full table
Quality of evidence
The results of GRADE on nosocomial infections showed that the quality of evidence were low or very low. The details can be found in the Supplement III (Table S4).
Full table
Discussion
Our rapid review identified a total of 40 studies. Low to very low-quality evidence indicated that the proportion of nosocomial infection among confirmed cases of COVID-19 was 44%, which is higher than for SARS but lower than for MERS. Most patients with COVID-19 and SARS infected in hospitals were medical staff, among whom nurses formed the largest group, followed by doctors. Both SARS and MERS outbreaks have been reported in hospitals, but we found no evidence of a COVID-19 outbreak.
SARS-CoV-2, the infectious agent causing COVID-19, is highly contagious, mainly spread by droplets and close contact. So far, a number of familial disease clusters have been reported, and some of the confirmed patients had been infected in healthcare facilities. As health care workers are in contact with a large number of suspected patients on a daily basis, strict precautions need to be taken to avoid outbreaks of infection in health care facilities. In the early stage of the epidemic, some hospitals, staff or publics did not have enough knowledge about the virus, leading to inadequate prevention and control measures, which may explain the reasons why the proportions of nosocomial infection are high in our study. The proportions may be higher than the real ones because the data of COVID-19 were from the early outbreak in Wuhan. When COVID-19 broke out in Wuhan at the beginning, medical resources were scarce, and various protective measures and management of hospitals were not in place, resulting in a high rate of nosocomial infections. Suspected patients did often not take any protection measures when they went to the hospital, which may have caused nosocomial infections and hospital outbreaks (19,20). A MERS study showed routine infection-prevention policies can greatly reduce nosocomial transmission of MERS (57). According to a report by the WHO, 20% of confirmed cases of SARS were among health care workers (58). Due to the rapidly evolving outbreak and spread of the disease, medical staff need to work in a state of high tension, but they should also protect themselves adequately and take the appropriate isolation measures to avoid cross infection in the hospital.
The high presence of the COVID-19 epidemic in the media is likely to improve the general public’s awareness. People with symptoms indicating a SARS-CoV-2 infection should take protective measures during the hospital or clinic visit, such as wearing a mask, minimizing the time of stay in the hospital, and if possible, making remote medical consultation in advance. Medical institutions should formulate sound infection prevention and control strategies, and strengthen the hospital's infection prevention and control efforts, such as the establishment of special departments for outpatients with fever, and a sound triage system: triage of early identification among suspected cases can avoid excessive gathering of patients in the hospital. Isolation wards should be established for suspected and confirmed patients needing treatment. In hospitals without single isolation wards or negative pressure isolation, indoor ventilation measures should be taken timely, and the management of patients should be standardized in these wards. Using adequate disinfection procedures can reduce the possibility of hospital transmission of the virus. During the epidemic, efforts should be made to publicize the knowledge of infection prevention and control, be alert to the possibility of the outbreak of nosocomial infection, and establish an early warning mechanism. Emergency plans or measures should be developed to deal with nosocomial infections.
Strengths and limitations
Our study included studies related to nosocomial infections among COVID-19, SARS and MERS patients. Our results can help the decision-making related to prevention, control and clinical management in hospitals. Some studies had missing data, and we used methods of meta-analyses of proportions to analyse those studies with available data, so the proportions estimated may not be accurate and similar to the actual data. Most of the results are based on low-quality research, so that the credibility of the results is low.
Conclusions
A large proportion of confirmed cases of COVID-19 were infected within healthcare facilities. Therefore, the patients who come to the hospital should do pay attention on personal protection. At the same time, medical institutions can reduce the spread of the virus through triage, and setting up separate fever clinic and isolation wards. Awareness of the disease needs to be improved among medical staff, so that they can protect themselves adequately and stop the spread of the virus within hospitals.
Supplementary I
Search strategy
PubMed
#1 “COVID-19”[Supplementary Concept]
#2 “Severe Acute Respiratory Syndrome Coronavirus 2”[Supplementary Concept]
#3 “Middle East Respiratory Syndrome Coronavirus”[Mesh]
#4 “Severe Acute Respiratory Syndrome”[Mesh]
#5 “SARS Virus”[Mesh]
#6 “COVID-19”[Title/Abstract]
#7 “SARS-COV-2”[Title/Abstract]
#8 “Novel coronavirus”[Title/Abstract]
#9 “2019-novel coronavirus”[Title/Abstract]
#10 “coronavirus disease-19”[Title/Abstract]
#11 “coronavirus disease 2019”[Title/Abstract]
#12 “COVID19”[Title/Abstract]
#13 “Novel CoV”[Title/Abstract]
#14 “2019-nCoV”[Title/Abstract]
#15 “2019-CoV”[Title/Abstract]
#16 “Middle East Respiratory Syndrome”[Title/Abstract]
#17 “MERS”[Title/Abstract]
#18 “MERS-CoV”[Title/Abstract]
#19 “Severe Acute Respiratory Syndrome”[Title/Abstract]
#20 “SARS”[Title/Abstract]
#21 “SARS-CoV”[Title/Abstract]
#22 “SARS-Related”[Title/Abstract]
#23 “SARS-Associated”[Title/Abstract]
#24 #1-#23/OR
#25 “Cross Infection”[MeSH Terms]
#26 “Cross Infection*”[Title/Abstract]
#27 “Healthcare Associated Infections*”[Title/Abstract]
#28 “Health Care Associated Infection*”[Title/Abstract]
#29 “Hospital Infection*”[Title/Abstract]
#30 “Nosocomial Infection*”[Title/Abstract]
#31 “hospital-related infection*”[Title/Abstract]
#32 “hospital-acquired infection*”[Title/Abstract]
#33 #25-#32/OR
#34 #24 AND #33
Embase
#1 ‘middle east respiratory syndrome coronavirus’/exp
#2 ‘severe acute respiratory syndrome’/exp
#3 ‘sars coronavirus’/exp
#4 ‘COVID-19’:ab,ti
#5 ‘SARS-COV-2’:ab,ti
#6 ‘novel coronavirus’:ab,ti
#7 ‘2019-novel coronavirus’:ab,ti
#8 ‘coronavirus disease-19’:ab,ti
#9 ‘coronavirus disease 2019’:ab,ti
#10 ‘COVID19’:ab,ti
#11 ‘novel cov’:ab,ti
#12 ‘2019-ncov’:ab,ti
#13 ‘2019-cov’:ab,ti
#14 ‘middle east respiratory syndrome’:ab,ti
#15 ‘middle east respiratory syndrome coronavirus’:ab,ti
#16 ‘mers’:ab,ti
#17 ‘mers-cov’:ab,ti
#18 ‘severe acute respiratory syndrome’:ab,ti
#19 ‘sars’:ab,ti
#20 ‘sars-cov’:ab,ti
#21 ‘sars-related’:ab,ti
#22 ‘sars-associated’:ab,ti
#23 #1-#22/OR
#24 ‘hospital infection*’:ab,ti
#25 ‘nosocomial infection*’:ab,ti
#26 ‘hospital-related infection*’:ab,ti
#27 ‘hospital-acquired infection*’:ab,ti
#28 ‘cross infection*’:ab,ti
#29 ‘healthcare associated infection*’:ab,ti
#30 ‘health care associated infection*’:ab,ti
#31 #24-#30/OR
#32 23 AND #31
Web of Science
#1 TOPIC: “COVID-19”
#2 TOPIC: “SARS-COV-2”
#3 TOPIC: “Novel coronavirus”
#4 TOPIC: “2019-novel coronavirus”
#5 TOPIC: “coronavirus disease-19”
#6 TOPIC: “coronavirus disease 2019”
#7 TOPIC: “COVID 19”
#8 TOPIC: “Novel CoV”
#9 TOPIC: “2019-nCoV”
#10 TOPIC: “2019-CoV”
#11 TOPIC: “Middle East Respiratory Syndrome”
#12 TOPIC: “MERS”
#13 TOPIC: “MERS-CoV”
#14 TOPIC: “Severe Acute Respiratory Syndrome”
#15 TOPIC: “SARS”
#16 TOPIC: “SARS-CoV”
#17 TOPIC: “SARS-Related”
#18 TOPIC: “SARS-Associated”
#19 #1-#18/OR
#20 TITLE: “Healthcare Associated Infection”
#21 TITLE: “Healthcare Associated Infections”
#22 TITLE: “Health Care Associated Infection”
#23 TITLE: “Health Care Associated Infections”
#24 TITLE: “Hospital Infection”
#25 TITLE: “Nosocomial Infection”
#26 TITLE: “Nosocomial Infections”
#27 TITLE: “Hospital Infections”
#28 TITLE: “hospital-related infection”
#29 TITLE: “hospital-acquired infection”
#30 TITLE: “Cross Infection”
#31 TITLE: “Cross Infections”
#32 #20-#31/OR
#33 #19 AND #32
Cochrane Library
#1 MeSH descriptor: [Middle East Respiratory Syndrome Coronavirus] explode all trees
#2 MeSH descriptor: [Severe Acute Respiratory Syndrome] explode all trees
#3 MeSH descriptor: [SARS Virus] explode all trees
#4 “COVID-19”:ti,ab,kw
#5 “SARS-COV-2”:ti,ab,kw
#6 “Novel coronavirus”:ti,ab,kw
#7 “2019-novel coronavirus” :ti,ab,kw
#8 “Novel CoV”:ti,ab,kw
#9 “2019-nCoV”:ti,ab,kw
#10 “2019-CoV”:ti,ab,kw
#11 “coronavirus disease-19”:ti,ab,kw
#12 “coronavirus disease 2019”:ti,ab,kw
#13 “COVID19”:ti,ab,kw
#14 “Middle East Respiratory Syndrome”:ti,ab,kw
#15 “MERS”:ti,ab,kw
#16 “MERS-CoV”:ti,ab,kw
#17 “Severe Acute Respiratory Syndrome”:ti,ab,kw
#18 “SARS”:ti,ab,kw
#19 “SARS-CoV”:ti,ab,kw
#20 “SARS-Related”:ti,ab,kw
#21 “SARS-Associated”:ti,ab,kw
#22 #1-#21/OR
#23 “hospital-related infection*”:ti,ab,kw
#24 “hospital-related infection*”:ti,ab,kw
#25 “cross infection*”:ti,ab,kw
#26 “healthcare associated infection*”:ti,ab,kw
#27 “health care associated infection*”:ti,ab,kw
#28 “hospital infection*”:ti,ab,kw
#29 “nosocomial infection*”:ti,ab,kw
#30 #23-#29/OR
#31 #22 AND #30
CNKI
#1 “新型冠状病毒”[主题]
#2 “COVID-19”[主题]
#3 “COVID 19”[主题]
#4 “2019-nCoV”[主题]
#5 “2019-CoV”[主题]
#6 “SARS-CoV-2”[主题]
#7 “中东呼吸综合征”[主题]
#8 “MERS”[主题]
#9 “MERS-CoV”[主题]
#10 “严重急性呼吸综合征”[主题]
#11 “SARS”[主题]
#12 #1-#11/ OR
#13 “医院相关感染” [主题]
#14 “医院获得性感染” [主题]
#15 “医疗机构相关感染” [主题]
#16 “院内感染” [主题]
#17 “交叉感染” [主题]
#18 #13-#17/ OR
#19 #12 AND #18
Wanfang
#1 “新型冠状病毒”[主题]
#2 “COVID-19”[主题]
#3 “COVID 19”[主题]
#4 “2019-nCoV”[主题]
#5 “2019-CoV”[主题]
#6 “SARS-CoV-2”[主题]
#7 “中东呼吸综合征”[主题]
#8 “MERS”[主题]
#9 “MERS-CoV”[主题]
#10 “严重急性呼吸综合征”[主题]
#11 “SARS”[主题]
#12 #1-#11/ OR
#13 “医院相关感染”)[主题]
#14 (“医院获得性感染”)[主题]
#15 (“医疗机构相关感染”)[主题]
#16 (“院内感染”)[主题]
#17 (“交叉感染”)[主题]
#18 #13-#17/OR
#19 #12 AND #18
CBM
#1 “新型冠状病毒”[常用字段:智能]
#2 “COVID-19”[常用字段:智能]
#3 “COVID 19”[常用字段:智能]
#4 “2019-nCoV”[常用字段:智能]
#5 “2019-CoV”[常用字段:智能]
#6 “SARS-CoV-2”[常用字段:智能]
#7 “中东呼吸综合征冠状病毒”[不加权:扩展]
#8 “中东呼吸综合征”[常用字段:智能]
#9 “MERS”[常用字段:智能]
#10 “MERS-CoV”[常用字段:智能]
#11 “严重急性呼吸综合征”[不加权:扩展]
#12 “SARS病毒”[不加权:扩展]
#13 “严重急性呼吸综合征”[常用字段:智能]
#14 “SARS”[常用字段:智能]
#15 #1-#14/OR
#16 “医院相关感染”[常用字段:智能]
#17 “医院获得性感染”[常用字段:智能]
#18 “医疗机构相关感染”[常用字段:智能]
#19 “交叉感染”[常用字段:智能]
#20 “院内感染"[常用字段:智能]
#21 #16-#20/OR
#22 #15 AND #21
Supplementary II
Risk of bias in the included studies
Supplementary III
Summary of findings
Acknowledgments
We thank Janne Estill, Institute of Global Health of University of Geneva for providing guidance and comments for our review. We thank all the authors for their wonderful collaboration.
Funding: This work was supported by grants from National Clinical Research Center for Child Health and Disorders (Children’s Hospital of Chongqing Medical University, Chongqing, China) (grant number NCRCCHD-2020-EP-01) to Enmei Liu; Special Fund for Key Research and Development Projects in Gansu Province in 2020, to Yaolong Chen; The fourth batch of “Special Project of Science and Technology for Emergency Response to COVID-19” of Chongqing Science and Technology Bureau, to Enmei Liu; Special funding for prevention and control of emergency of COVID-19 from Key Laboratory of Evidence Based Medicine and Knowledge Translation of Gansu Province (grant number No. GSEBMKT-2020YJ01), to YC; The Fundamental Research Funds for the Central Universities (lzujbky-2020-sp14), to YC and KY.
Footnote
Reporting Checklist: The authors have completed the PRISMA reporting checklist. Available at http://dx.doi.org/10.21037/atm-20-3324
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/atm-20-3324). MSL serves as the unpaid editorial board member of Annals of Translational Medicine from Nov 2019 to Oct 2021. The other authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020;395:497-506. [Crossref] [PubMed]
- National Health Commission & National Administration of Traditional Chinese Medicine. Diagnosis and Treatment Protocol for Novel Coronavirus Pneumonia (Trial Version 7) [J/OL] . Chin Med J,2020,133 (2020-03-13). Available online: http://rs.yiigle.com/yufabiao/1184760.htm. doi: 10.3760/cma.j.issn.0366-6999.2020.0027. [Crossref]
- Liu Y, Gayle AA, Wilder-Smith A, et al. The reproductive number of COVID-19 is higher compared to SARS coronavirus. J Travel Med 2020;27:taaa021.
- Khan S, Ali A, Siddique R, et al. Novel coronavirus is putting the whole world on alert. J Hosp Infect 2020;104:252-3. [Crossref] [PubMed]
- WHO Director-General's opening remarks at the media briefing on COVID-19 - 11 March 2020[Internet]. World Health Organization;c2020 [cited 2020 Apr 13]. Available online: https://www.who.int/dg/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19---11-march-2020
- Coronavirus disease (COVID-19) outbreak situation Data as reported by national authorities by 7 April 2020, 08:00 GMT+8[Internet]. World Health Organization; c2020 [cited 2020 Apr 13]. Available online: https://www.who.int/emergencies/diseases/novel-coronavirus-2019
- Chan JF, Yuan SF, Kok KH, et al. A familial cluster of pneumonia associated with the 2019 novel coronavirus indicating person-to-person transmission: a study of a family cluster. Lancet 2020;395:514-23. [Crossref] [PubMed]
- Wang D, Hu B, Hu C, et al. Clinical Characteristics of 138 Hospitalized Patients With 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China. JAMA 2020;323:1061-9. [Crossref] [PubMed]
- The Novel Coronavirus Pneumonia Emergency Response Epidemiology Team. Vital surveillances: the epidemiological characteristics of an outbreak of 2019 novel coronavirus diseases (COVID-19)—China, 2020. China CDC Weekly. 2020;2:113-22. Available online: http://weekly.chinacdc.cn/en/article/id/e53946e2-c6c4-41e9-9a9b-fea8db1a8f51
- Li L, Tian JH, Tian HL, et al. Network meta-analyses could be improved by searching more sources and by involving a librarian. J Clin Epidemiol 2014;67:1001-7. [Crossref] [PubMed]
- Moher D, Liberati A, Tetzlaff J, et al. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med 2009;6:e1000097. [Crossref] [PubMed]
- Wells G, Shea B, O'Connell D, et al. NewCastle-Ottawa Quality Assessment Scale --Cohort Studies. [cited 2020 Mar 31]. Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp
- Rostom A, Dubé C, Cranney A, et al. Celiac Disease. Rockville (MD): Agency for Healthcare Research and Quality (US); 2004 Sep. Evidence Reports/Technology Assessments, No. 104. Appendix D. Quality Assessment Forms. Available online: https://www.ncbi.nlm.nih.gov/books/NBK35156/
- National Institute for Health and Care Excellence 2003 [cited 2020 Apr 13] Available online: https://www.nice.org.uk/guidance/cg3/documents/appendix-4-quality-of-case-series-form2
- Norris SL, Meerpohl JJ, Akl EA, et al. The skills and experience of GRADE methodologists can be assessed with a simple tool. J Clin Epidemiol 2016;79:150-8.e1. [Crossref] [PubMed]
- Guyatt GH, Oxman AD, Vist GE, et al. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ 2008;336:924-6. [Crossref] [PubMed]
- Ge L, Tian J, Li Y, et al. Association between prospective registration and overall reporting and methodological quality of systematic reviews: a meta-epidemiological study. J Clin Epidemiol 2018;93:45-55. [Crossref] [PubMed]
- Wang Q, Kuang W, Ping W, et al. Prevention and treatment of cross infection of novel coronavirus pneumonia in thoracic surgery ward. Chin J Thoracic Cardiovasc Surg 2020;27:371-5.
- Jiang W, Lu Z, Shen X, et al. Clinical practice of prevention and treatment of novel coronavirus infection in the medical personnel and surgical patients in the Department of Thoracic Surgery of Hospitals in Wuhan. Chin J Thoracic Cardiovasc Surg 2020;27:364-70.
- Shen Y, Ke C, Yang C, et al. A case-control study on 2019-nCov infection-related factors among medical staff in Wuhan Tongji Hospital. Chin J Nosocomiology 2020;30:1157-60.
- Bi X, Zhou F, Zhang K. A cluster of cases of infectious SARS in a hospital. Chin J Resp Crit Care Med 2003;2:161-3.
- Dai L, Gao Y, Zeng W. Epidemiological investigation of nosocomial infection of SARS in a Chinese medicine hospital in Guangdong Province. Chin J Public Health Management 2004;20:242.
- Zou Q, Lin WS, Du L. An investigation on nosocomial infection with severe acute respiratory syndrome in health-care workers at 13 key hospitals in Guangdong Province. Zhonghua Liu Xing Bing Xue Za Zhi 2004;38:87-9. [PubMed]
- Wang M, Du L, Zhou D, et al. Study on the epidemiology and measures for control on severe acute respiratory syndrome in Guangzhou city. Zhonghua Liu Xing Bing Xue Za Zhi 2003;24:353-7. [PubMed]
- Gao L, Zhang X, Yin W. Investigation and analysis of nosocomial infection of SARS among medical staff in a hospital in Guangzhou. South Chin J Prevent Med 2003;29:19-20.
- Lin W, Yin W, Zhou Q. Analysis of the effect of the preventive measures against SARS infection in a hospital in Guangzhou. South Chin J Prevent Med 2003;29:17-8.
- Xu H, Wang M, Liu Y. SARS outbreak and control in hospital health care workers in Guangzhou. South Chin J Prevent Med 2003;29:16-8.
- Gao X, Chang A, Yang Z. Analysis of communicated severe acute respiratory syndrome in staff of a general hospital. J Logistics U Capf 2003;12:424-7.
- Yuan J, Hu Y, Jiang X. Epidemiological Analysis on Severe Acute Respiratory Syndrome in Shenzhen city. J Tropical Med 2003;3:423-4, 83.
- Wang JX, Feng H, Liu D. Epidemiological characteristics of severe acute respiratory syndrome in Tianjin and the assessment of effectiveness on measures of control. Zhonghua Liu Xing Bing Xue Za Zhi 2003;24:565-9. [PubMed]
- Wang G, Zhao W, Zhang F. The causes and control strategies of epidemic severe acute respiratory syndrome in hospitals. Chin J Infect Control 2003;2:165-7.
- Wu J, Li L, Xu Y. Role of Hospital Infection Control System in Decreasing SARS Infection among Medical Staff. Chin J Nosocomiology 2004;14:903-5.
- Huang L, Zhang S, Yin C. Analysing Causes and Preventive Measures of Occurring SARS among Medical Workers. Chin J Nosocomiology 2003;13:1050-2.
- Li G, Gu Y, Liang H. Epidemiological analysis on SARS cases in Zhongshan. South Chin J Prevent Med 2003;29:42-3.
- Fei Y, He Y, Liu D. Study and evaluation on the measures of prevention and control in SARS hospital infection. J Chin PLA Postgraduate Medical School 2003;24:304-6.
- Lu H, Huo N, Xu X. The epidemiological characteristics of patients with Severe Acute Respiratory Syndrome (SARS). Beijing Da Xue Xue Bao Yi Xue Ban 2003;35:8-11. [PubMed]
- He X,Liu Z, Ma Y, et al. Epidemic characteristics of SARS in Beijing in 2003. Xiangshan Science Meeting, 2003.
- Ho AS, Sung JJ, Chan-Yeung M. An outbreak of severe acute respiratory syndrome among hospital workers in a community hospital in Hong Kong. Ann Intern Med 2003;139:564-7. [Crossref] [PubMed]
- Li L, Cheng S, Gu J. Analysis of medical workers suffered from SARS. Beijing Da Xue Xue Bao Yi Xue Ban 2003;35:51-4. [PubMed]
- Fowler RA, Lapinsky SE, Hallett D, et al. Critically Ill Patients with Severe Acute Respiratory Syndrome. JAMA 2003;290:367-73. [Crossref] [PubMed]
- Varia M, Wilson S, Sarwal S, et al. Investigation of a nosocomial outbreak of severe acute respiratory syndrome (SARS) in Toronto, Canada. CMAJ 2003;169:285-92. [PubMed]
- Lau JTF, Yang X, Leung PC, et al. SARS in three categories of hospital workers, Hong Kong. Emerg Infect Dis 2004;10:1399-404. [Crossref] [PubMed]
- Zhou Y, Chen S, Gao Y. Protective Measures to Medical Personnel and Their Effectiveness in SARS Prevention During the SARS Outbreak in Guangdong Province. Chin J Nosocomiology 2004;14:433-5.
- Chen M, Leo YS, Ang B, et al. The outbreak of SARS at Tan Tock Seng Hospital--relating epidemiology to control. Ann Acad Med Singapore. 2006;35:317-25. [PubMed]
- Cooper BS, Fang LQ, Zhou JP, et al. Transmission of SARS in three Chinese hospitals. Trop Med Int Health 2009;14 Suppl 1:71-8. [Crossref] [PubMed]
- Oboho IK, Tomczyk SM, Al-Asmari AM, et al. 2014 MERS-CoV outbreak in Jeddah-a link to health care facilities. N Engl J Med 2015;372:846-54. [Crossref] [PubMed]
- Xiang N, Lin D, An G. Epidemiological characteristics of Middle East Respiratory Syndrome outbreak in the Republic of Korea, 2015. Zhonghua Liu Xing Bing Xue Za Zhi 2015;36:836-41. [PubMed]
- Assiri A, McGeer A, Perl TM, et al. Hospital outbreak of Middle East respiratory syndrome coronavirus. N Engl J Med 2013;369:407-16. [Crossref] [PubMed]
- Alenazi TH, Al Arbash H, El-Saed A, et al. Identified Transmission Dynamics of Middle East Respiratory Syndrome Coronavirus Infection During an Outbreak: Implications of an Overcrowded Emergency Department. Clin Infect Dis 2017;65:675-9. [Crossref] [PubMed]
- Memish ZA, Al-Tawfiq JA, Alhakeem RF, et al. Middle East respiratory syndrome coronavirus (MERS-CoV): A cluster analysis with implications for global management of suspected cases. Travel Med Infect Dis 2015;13:311-4. [Crossref] [PubMed]
- Park SH, Kim Y, Jung Y, et al. Outbreaks of middle east respiratory syndrome in two hospitals initiated by a single patient in Daejeon, South Korea. Infect Chemother 2016;48:99-107. [Crossref] [PubMed]
- Al-Dorzi HM, Aldawood AS, Khan R, et al. The critical care response to a hospital outbreak of Middle East respiratory syndrome coronavirus (MERS-CoV) infection: an observational study. Ann Intensive Care 2016;6:101. [Crossref] [PubMed]
- Hunter JC, Nguyen D, Aden B, et al. Transmission of Middle East Respiratory Syndrome Coronavirus Infections in Healthcare Settings, Abu Dhabi. Emerg Infect Dis 2016;22:647-56. [Crossref] [PubMed]
- Amer H, Alqahtani AS, Alzoman H, et al. Unusual presentation of Middle East respiratory syndrome coronavirus leading to a large outbreak in Riyadh during 2017. Am J Infect Control 2018;46:1022-5. [Crossref] [PubMed]
- Cho SY, Kang JM, Ha YE, et al. MERS-CoV outbreak following a single patient exposure in an emergency room in South Korea: an epidemiological outbreak study. Lancet 2016;388:994-1001. [Crossref] [PubMed]
- Hijawi B, Abdallat M, Sayaydeh A, et al. Novel coronavirus infections in Jordan, April 2012: epidemiological findings from a retrospective investigation. East Mediterr Health J 2013;19 Suppl 1:S12-8. [Crossref] [PubMed]
- Ki HK, Han SK, Son JS, et al. Risk of transmission via medical employees and importance of routine infection-prevention policy in a nosocomial outbreak of Middle East respiratory syndrome (MERS): a descriptive analysis from a tertiary care hospital in South Korea. BMC Pulm Med 2019;19:190. [Crossref] [PubMed]
- Summary table of SARS cases by country, 1 November 2002 - 7 August 2003[Internet]. World Health Organization; c2020 [cited 2020 Apr 13]. Available online: https://www.who.int/csr/sars/country/country2003_08_15.pdf?ua=1