The epidemiologic and clinical features of suspected and confirmed cases of imported 2019 novel coronavirus pneumonia in north Shanghai, China
Introduction
In December 2019, a series of pneumonia cases of unknown causes emerged in Wuhan, Hubei Province of China (1-3). A novel coronavirus was identified by the Chinese Center for Disease Control and Prevention (CDC) from the lower respiratory tract sample of a patient, which was subsequently named corona virus disease 2019 (COVID-19) by the WHO in February 11th, 2020 (4).
Some patients with COVID-19 infection have developed severe pneumonia, pulmonary edema, acute respiratory distress syndrome (ARDS) and multiple organ failure (MOF), or even died. Up to now, the exact transmission route of COVID-19 remains unknown, but evidence of human-to-human transmission or affected health-care workers (HCWs) has been certificated. Other provinces in China, Thailand, Japan, South Korea, and the USA have also several exported cases (5-8). Shanghai is the largest base of Chinese industrial technology, and the largest commercial and financial center. At present, there is little information regarding the epidemiological and clinical features of confirmed and suspected COVID-19 cases.
In this study, we intend to compare the epidemiological, clinical, laboratory and radiological characteristics, treatment and outcomes between patients with imported confirmed COVID-19 infection and those with suspected COVID-19 infection through analyzing the cases detected in north Shanghai, China. We present the following article in accordance with STROBE reporting checklist (available at http://dx.doi.org/10.21037/atm-20-2119).
Methods
Study design and participants
Included in this retrospective two-center study were patients were identified as having COVID-19 pneumonia between January 1 and February 15, 2020 at Shanghai Tenth People’s Hospital and Shibei Hospital in Shanghai, China. According to Shanghai Municipal Center for Disease Control & Prevention (CDC), all patients who were confirmed as having COVID-19 pneumonia must be transferred to Shanghai Public Health Clinical Center for uniform isolation and treatment, and therefore this study did not discuss the detail treatment of those confirmed cases. The diagnosis standard of suspected cases is issued according to diagnosis and treatment of novel coronavirus pneumonia (Trial version fifth) by National Health and Health Commission of the people’s Republic of China in February 5th, 2020 (9). The study protocol was approved by the ethics committee of the said two hospitals (No. SHSY-IEC-4.1/20-21/01), and informed consent was obtained from the patients or their families before enrolment.
Procedures
Data about the epidemiological and clinical features were obtained from the medical records of the patients. Throat swab and sputum specimens were obtained from all patients and sent for COVID-19 detection at the laboratories of Shanghai Municipal CDC by real-time PCR (RT-PCR) as described (2,3). Other respiratory viruses including influenza A virus (H1N1, H3N2 and H7N9), influenza B virus, SARS coronavirus (SARS-CoV), and MERS coronavirus (MERS-CoV) were also detected by RT-PCR.
Outcomes
The epidemiological features, patient demographics, symptoms and signs at admission, comorbidity, laboratory findings, chest CT findings and clinical outcomes of the suspected and confirmed imported cases were analyzed and compared.
Statistical analysis
Statistical analysis was performed using SPSS version 20.0. The results are showed as the mean ± standard error (SE) for at least three independent experiments. Student’s t-test and one-way ANOVA were applied to the experiment results. Differences were considered statistically significant when P<0.05.
Results
Factors associated with confirmed and suspected group
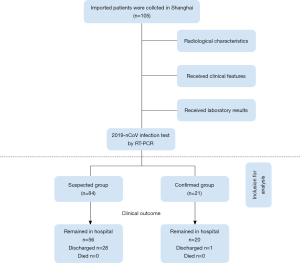
By February 15, 2020, 105 patients had been included in this study, of whom 21 patients were identified as having laboratory-confirmed COVID-19 infection, including a mother and a son, and a couple (Figure 1). The median time from symptom onset to diagnosis, from hospital admission to diagnosis, and diagnosis to hospital transfer in these confirmed cases was 145.64 h (IQR 21–441 h), 22 h (IQR 16–33 h), and 14.79 h (IQR 1–52 h) respectively.
Fourteen (66.67%) of the 21 confirmed patients were older than 51 (range, 51–60) years, while 50 (59.5%) of the 84 suspected patients were younger than 40 years, including 27 (32%) younger than 30 years (Figure 2A). More elderly people were diagnosed with COVID-19, while more younger people were included in the suspected group (Figure 2B). Thirteen (61.9%) of 21 confirmed cases were distributed in Jing’an District of North shanghai (Figure 2C).

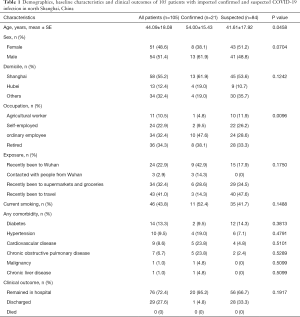
The baseline characteristics of the patients are shown in Table 1. Most confirmed patients were men (61.9%, 13/21) with a mean age of 54.00±15.43 years, and about 50% of them had underlying diseases, including diabetes (9.5%, 2/21), hypertension (19%, 4/21), chronic obstructive pulmonary disease (COPD) (23.8%, 5/21), and cardiovascular disease (CD) (23.8%, 5/21). In addition, 10 (47.6%) of the 21 patients with confirmed COVID-19 infection were ordinary employees and had recently been to Wuhan or had close contacts with people from Wuhan (57.2%, 12/21).
Full table
Forty-three (51.2%) of the 84 suspected patients were women with a mean age of 41.61±17.92 years, and fewer of them had underlying diseases, including diabetes (14.3%, 12/84), hypertension (7.1%, 6/84), COPD (4.8%, 4/84), and CD (2.4%, 2/84). Twenty-eight (33.3%) of the suspected patients were retired, and had recently been to supermarkets and groceries, or had a recent history of traveling abroad or to other cities of China (82.1%, 69/84). There were significant differences in age, occupation and exposure between the two groups (P<0.005), but there was no significant difference in domiciles and comorbidity between the two groups (P>0.05).
Comparison of the clinical characteristics and laboratory findings between the confirmed and suspected groups
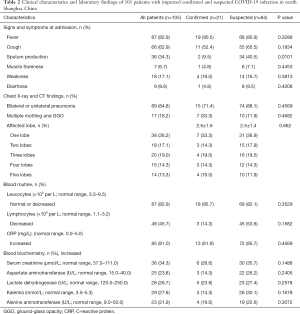
The common onset symptoms at admission in the confirmed group were fever (90.5%, 19/21), cough (52.4%, 11/21), weakness (19%, 4/21), muscle soreness (4.8%, 1/21), and diarrhea (4.8%, 1.21). The common symptoms in the suspected group were fever (80.9%, 68/84), cough (65.5%, 55/84), and weakness (16.7%, 14/84). The common onset symptoms were similar between the two groups, but productive cough at admission was more frequently reported in suspected patients that that in confirmed patients (40.5%, 34/84 vs. 9.5%, 2/21, P=0.010) (Table 2).
Full table
Leucocyte count at admission was nearly normal in both confirmed group (85.7%, 18/21) and suspected group (82.1%, 69/84). Lymphocyte count was also nearly normal in both groups (53.6%, 45/84 vs. 14.3%, 3/21). C-reactive protein (CRP) was above the normal range in 13 (61.9%) of the 21 confirmed patients and 72 (85.7%) of the 84 suspected patients. There were no significant differences in blood routine at admission between the two groups (P>0.05) (Table 2).
Varying degrees of liver function abnormality was detected in 7 (34%) of the confirmed group and 48 (56.1%) of the suspected group, as represented by elevation of alanine aminotransferase (ALT) or aspartate aminotransferase (AST). Abnormal kalemia was detected in 3 (14.3%) of the confirmed patients and 26 (30.1%) of the suspected patients. Varying degrees of renal function impairment as represented by blood urea nitrogen (BUN) elevation were detected in 6 (28.6%) of the confirmed patients and 30 (35.7%) of the suspected patients. There were no significant differences in blood biochemistry at admission between the two groups (P>0.05) (Table 2).
Comparison of CT findings between the confirmed and suspected groups
CT images of 105 imported patients were collected. The mean number of affected lobes in confirmed group was 2.7±1.6. Seven (33.3%) patients had one affected lobe, three (14.3%) patients had two affected lobes, four (19%) patients had three affected lobes, three (14.3%) patients had four affected lobes, and four (19%) patients had 5 affected lobes. CT imaging showed bilateral or unilateral pneumonia in 15 (71.4%) patients of the confirmed group and 74 (88.1%) patients of the suspected group, and multiple mottling and ground-glass opacity (GGO) was detected in 7 (33.3%) patients of the confirmed group and 10 (11.9%) patients of the suspected group (Table 2; Figure S1).
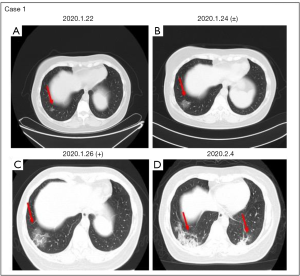
Case 1 in the confirmed group
A 47-year-old female patient who had a 7-day history of travelling to Wuhan complained of fever (37.6 °C) for one day with mild cough. CT scan revealed a small inflamed nodule in the lower lobe of the right lung, with no obvious exudation found in other lung fields (Figure 3A). Her blood routine results were normal. After two days, a flocculent fuzzy exudation shadow was found in the basal segment of the right inferior lung lobe. At the same time, RT-PCR revealed a weakly positive result for COVID-19 (Figure 3B). Repeated CT and RT-PCR assay showed high-density shadows in both lungs and a positive COVID-19 result (Figure 3C,D).
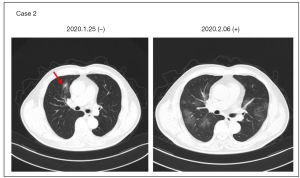
Case 2 in the confirmed group
A male patient who returned to Shanghai from a Wuhan trip on January 20, 2020 complained of fever for two days without other symptoms. Chest CT on 2020-01-25 showed exudation and nodular shadows near the bronchus of the upper lobe of the right lung (Figure 4A). COVID-19 tests on 2020-01-25 and 01-26 were both negative. As fever persisted for a week, he went to the hospital again and underwent a third CT scan and RT-PCR. CT showed multiple exudative GGO shadows in both lungs (Figure 4B), and RT-PCR showed a positive result for COVID-19, and a further chest CT scan showed progression of the condition.
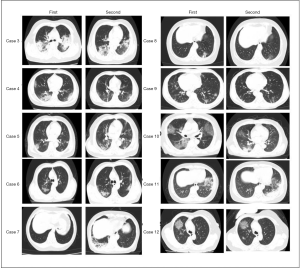
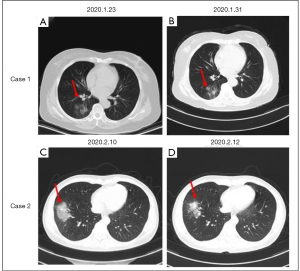
There was no significant difference in CT presentation between the confirmed and suspected groups at the time of admission (P>0.05), but the disease progressed faster and more quickly in patients of the confirmed group (76.2%, 16/21), including single ground glass nodules increased, enlarged and consolidated; fibrous stripe enlarged, while solid nodules increased and enlarged (Figure S1), while no significant change was noticed in the suspected group (Figure 5, Cases 1 and 2).
Comparison of the clinical outcomes between the confirmed and suspected groups
Bacteria and fungi, as well as nucleic acid of influenza virus A and B were detected in all patients of both groups. Only one patient in confirmed group combined influenza A was admitted immediately to Shanghai Public Health Clinical Center and received intubated ventilator assisted breathing therapy (Figure S1, Case 3). Now the patient has been discharged after 14-day intensive treatment.
All confirmed patients were mainly treated with traditional Chinese medicines, lopinavir and ritonavir. Most suspected patients were given antibiotic treatment. The antibiotics used generally covered common pathogens and some atypical pathogens. Only one confirmed patient with influenza A received antiviral treatment, including oseltamivir and ganciclovir. No glucocorticoid was used in any of the confirmed patients.
By the end of February 15, 29 (27.6%) of the 105 included patients had been clinically cured and discharged, and no patient had died. The remaining patients are still retained in the hospital (Table 1). No severe or critical case was found in all patients with imported COVID-19 infection.
Discussion
This is a descriptive study on the epidemiological and clinical features of 105 patients with confirmed or suspected imported COVID-19 infection in north Shanghai, China. It was found in our study that the overall incubation period of the COVID-19 imported patients in our series was longer than that in Wuhan. In additional, the mean number of affected lobes in imported patient was less as compared to Wuhan case. The disease onset of imported COVID-19 infection was occult, and the clinical symptoms were usually mild, mostly presenting as low fever, fatigue, light cough, and mild dyspnea.
Human coronavirus is one of the main pathogens of respiratory infection. SARS-CoV and MERS-CoV are two highly pathogenic viruses (10,11). SARS-CoV outbreak spread to 29 countries globally during 2002 and 2003. MERS-CoV emerged in Middle Eastern countries in 2012 (12,13). The sequence of COVID-19 is relatively different from the other coronavirus subtypes but can be classified as beta-coronavirus. There is evidence of human-to-human transmission (14). The mortality of SARS-CoV and MERS-CoV was reported to be more than 10% (15) and 35% (16) respectively. The COVID-19 infection reported in China is currently believed to originate from bats, causing a mortality of 11–15% (3). In summary, an outbreak of novel coronavirus is ongoing in Wuhan, China in the winter of 2019–2020. Wuhan is a rapidly nourishing capital city of Hubei Province of China and the traffic hub of central China. In our study, we report here a cohort of 21 patients with laboratory-confirmed COVID-19 infection and 84 suspected patients. The 21 confirmed patients including a mother and a son, and a couple. We found that the incubation period of the imported cases was 17.6 days (IQR 5–34 days), the longest incubation period being 34 days. The incubation period of these imported confirmed cases in Shanghai is obviously longer than that in Wuhan, and the possible reason is that the imported confirmed patients in our series had fewer underlying diseases (diabetes, hypertension, COPD, and CD). The patients with confirmed COVID-19 infection ranged in age from 51 to 60 years with a mean of 54.00±15.43 years, and most of the patients with suspected COVID-19 infection were younger than 30 years. In addition, most of the confirmed patients were ordinary employees and had a recent history of traveling to Wuhan or had close contacts with people from Wuhan. Of the 84 suspected patients, about 30% were retired, and most had recently been to supermarkets and groceries, or had a recent history of traveling abroad or to other cities of China. The clinical features of confirmed and suspected patients were similar. Fever and dry cough were the common onset symptoms in most of the imported cases in our series, and productive cough was mainly observed in patients of the suspected group. Chen et al observed a greater number of men than women in their 99 cases of COVID-19 infection (17), which is similar to the finding in our series. MERS-CoV and SARS-CoV have also been found to infect more men than women (18,19). These findings suggest that COVID-19 is more likely to infect older adult males.
As most of the imported patients in our series visited their doctors within one or two days after onset of the symptoms, the results of their laboratory tests showed a slight decrease leukocyte count and a slight increase in the monocyte count. The cause of abnormal leukocyte is that COVID-19 might mainly act on lymphocytes, especially T lymphocytes (17), and the virus particles spread via the respiratory mucosa and infect other cells, causing a cytokine storm in the body and producing a series of immune responses. Some patients rapidly progressed to ARDS and septic shock, eventually leading to MOF. So, the absolute value of leukocyte count in imported patients, which can be used as a reference index for clinical diagnosis of new coronavirus infection.
The imaging findings of the confirmed patients in our series are not typical, which is different from the Wuhan cases. Firstly, the mean number of affected lobes in imported group CT image was 2.7±1.6, and more than 50% imported patients (53.3%, 56/105) had one and two affected lobes. But, Pan et al. found that mean number of affected lobes in Wuhan infected patients was 3.3±1.8, and most patients had 5 affected lobes (20). The imported patient’s CT image affected less lobes as compared to Wuhan case. Secondly, Slight subpleural exudative lesions or multiple mild GGO exudative shadows are the main imaging presentations of the patients in our series. Three or five days later, typical lesions (bilateral or unilateral pneumonia) appeared on CT imaging, as represented by multiple GGO shadows or interstitial changes under the pleura (Figure S1). Owing to early detection and early treatment, no severe or critical cases were found in our confirmed cases, and these imaging lesions were absorbed quickly after appropriate treatment.
Although the suspected imported cases in our series met the diagnosis and treatment of novel coronavirus pneumonia (the trial version fifth) by China’s National Health Commission in February 5th, 2020 (9). the results of two consecutive PCR assays with a 24-h interval were both negative. The imaging features of the suspected cases are GGO exudation near the segmental bronchi, which then extended father away and fused into pieces. After short-term treatment, progress was halted or slowed down, and the lesions were even absorbed in some cases (Figure 5). We postulate that these suspected cases might be caused by other viruses or mycoplasma.
In one patient of the confirmed group in our series, RT-PCR was negative in the initial two consecutive assays, but the focus progressed rapidly on the CT reexamination image and the PCR test result turned positive, suggesting that for cases with typical clinical manifestations and typical image manifestations, isolation should not be removed too early after two consecutive PCR assays and CT should be reexamined within a short period. If the image performance progresses rapidly, PCR assay should be repeated two or more times. The false-negative result of PCR assay may be caused by the small amount of virus in the early stage of the disease, delayed submission of the PCR sample, unqualified sampling, or the false-negative rate of the reagent itself.
Most of the imported COVID-19 cases in north Shanghai are symptomatically mild with normal blood routine and biochemistry at admission and the patients had fewer underlying diseases and complications as compared the Wuhan cases. No corticosteroids were used in any of our 21 confirmed cases, knowing that the WHO interim guidance does not advocate systemic use of corticosteroids (21). But as corticosteroids were commonly used in the treatment of severe cases of SARS and MERS infection (22,23), further evidence is urgently required to verify whether systemic corticosteroid treatment is beneficial or harmful to patients infected with COVID-19. COVID-19 is a newly emerging virus, for which no effective treatment is available at present, and therefore antiviral treatment for previous coronavirus infections has been adopted as an interim therapy.
This study has some strengths and limitations. First, only 21 confirmed COVID-19 cases were included. But to the best of our knowledge, this is the first study to compare the epidemiological, clinical, laboratory and imaging differences between confirmed and suspected cases of COVID-19. Second, we were unable to provide data about recovery and the psychological status of the included patients because all confirmed cases had to be transferred to Shanghai Public Health Clinical Center for specific isolation and treatment. However, our data may help get a more comprehensive understanding about the epidemiological, clinical, laboratory and imaging features of imported cases of COVID-19 infection.
Conclusions
In conclusion, the incubation period of the imported cases of confirmed COVID-19 infection in Shanghai is longer than that in Wuhan, China. It mainly infected retired elderly men. The disease onset in patients with imported confirmed COVID-19 infection was occult. The clinical symptoms were generally mild, mainly presenting as low fever, fatigue, light cough and mild dyspnea. A good clinical outcome of COVID-19 pneumonia can be achieved by early diagnosis and appropriate treatment.
Acknowledgments
Funding: None.
Footnote
Reporting checklist: The authors have completed the STROBE reporting checklist (available at http://dx.doi.org/10.21037/atm-20-2119)
Data Sharing Statement: Available at http://dx.doi.org/10.21037/atm-20-2119
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/atm-20-2119). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was approved by Shanghai Tenth People’s Hospital and Shibei Hospital of Jing’an District (Shanghai, China) (No. SHSY-IEC-4.1/20-21/01). Written informed consent was obtained from the patient for publication of this study and any accompanying images.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Lu H, Stratton CW, Tang YW. Outbreak of pneumonia of unknown etiology in Wuhan China: the mystery and the miracle. J Med Virol 2020;92:401-2. [Crossref] [PubMed]
- Hui DS, I, Azhar E, Madani TA, et al. The continuing 2019nCoV epidemic threat of novel coronaviruses to global health the latest 2019 novel coronavirus outbreak in Wuhan, China. Int J Infect Dis 2020;91:264-6. [Crossref] [PubMed]
- Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020;395:497-506. [Crossref] [PubMed]
- WHO. Novel coronavirus–China. Jan 12, 2020. Available online: (accessed Jan 19, 2020).http://www.who.int/csr/don/12-january-2020-novel-coronavirus-china/en/
- WHO. Novel coronavirus–Thailand (ex-China). Jan 14, 2020. (accessed Jan 19, 2020).http://www.who.int/csr/don/14-january-2020-novel-coronavirus-thailand/en/
- WHO. Novel coronavirus–Japan (ex-China). Jan 17, 2020. Available online: (accessed Jan 19, 2020).http://www.who.int/csr/don/17-january-2020-novel-coronavirus-japan-ex-china/en/
- WHO. Novel coronavirus–Republic of Korea (ex-China). Jan 21, 2020. Available online: (accessed Jan 23, 2020).http://www.who.int/csr/don/21-january-2020-novel-coronavirus-republic-of-korea-ex-china/en/
- CDC. First travel-related case of 2019 novel coronavirus detected in United States. Jan 21, 2020. Available online: (accessed Jan 23, 2020).https://www.cdc.gov/media/releases/2020/p0121-novel-coronavirus-travel-case.html
- China, National Health Commission, Diagnosis and treatment of novel coronavirus pneumonia (trial version fifth) 2020.2.5. Available online: http://so.kaipuyun.cn/s?token=9052&siteCode=bm24000006&qt
- Hu B, Zeng LP, Yang XL, et al. Discovery of a rich gene pool of bat SARS -related coronaviruses provides new insights into the origin of SARS coronavirus. PLoS Pathog 2017;13:e1006698. [Crossref] [PubMed]
- Song HD, Tu CC, Zhang GW, et al. Cross-host evolution of severe acute respiratory syndrome coronavirus in palm civet and human. Proc Natl Acad Sci USA 2005;102:2430-35. [Crossref] [PubMed]
- Haagmans BL, Al Dhahiry SH, Reusken CB, et al. Middle East respiratory syndrome coronavirus in dromedary camels: an outbreak investigation. Lancet Infect Dis 2014;14:140-5. [Crossref] [PubMed]
- Azhar EI, El-Kafrawy SA, Farraj SA, et al. Evidence for camel-to-human transmission of MERS coronavirus. N Engl J Med 2014;370:2499-505. [Crossref] [PubMed]
- Perlman S, Netland J. Coronaviruses post-SARS: update on replication and pathogenesis. Nat Rev Microbiol 2009;7:439-50. [Crossref] [PubMed]
- Yin Y, Wunderink RG. MERS, SARS and other coronaviruses as causes of pneumonia. Respirology 2018;23:130-7. [Crossref] [PubMed]
- Song Z, Xu Y, Bao L, et al. From SARS to MERS, thrusting coronaviruses into the spotlight. Viruses 2019;11:59. [Crossref] [PubMed]
- Chen N, Zhou M, Dong X, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet 2020;395:507-13. [Crossref] [PubMed]
- Badawi A, Ryoo SG. Prevalence of comorbidities in the Middle East respiratory syndrome coronavirus (MERS-CoV): a systematic review and meta-analysis. Int J Infect Dis 2016;49:129-33. [Crossref] [PubMed]
- Channappanavar R, Fett C, Mack M, et al. Sex-based differences in susceptibility to severe acute respiratory syndrome coronavirus infection. J Immunol 2017;198:4046-53. [Crossref] [PubMed]
- Pan Y, Guan H, Zhou S, et al. Initial CT findings and temporal changes in patients with the novel coronavirus pneumonia (2019-nCoV): a study of 63 patients in Wuhan, China. Eur Radiol 2020. [Crossref] [PubMed]
- Stockman LJ, Bellamy R, Garner P. SARS: systematic review of treatment effects. PLoS Med 2006;3:e343. [Crossref] [PubMed]
- Lansbury L, Rodrigo C, Leonardi-Bee J, et al. Corticosteroids as adjunctive therapy in the treatment of in uenza. Cochrane Database Syst Rev 2019;2:CD010406. [PubMed]
- Arabi YM, Mandourah Y, Al-Hameed F, et al. Corticosteroid therapy for critically ill patients with Middle East respiratory syndrome. Am J Respir Crit Care Med 2018;197:757-67. [Crossref] [PubMed]