Chest drainage systems in use
Introduction
Pressure around the lungs is lower than atmospheric pressure outside the body.
The aims for an adequate chest drainage system to be fulfilled are: (I) remove fluid & air as promptly as possible; (II) prevent drained air & fluid from returning to the pleural space, restore negative pressure in the pleural space to re-expand the lung. Thus, a drainage device must: (I) allow air and fluid to leave the chest; (II) contain a one-way valve to prevent air & fluid returning to the chest; (III) have design so that the device is below the level of the chest tube for gravity drainage. An underwater seal chest drainage system is used to restore proper air pressure to the lungs, re-inflate a collapsed lung as well as remove blood and other fluids. The system is a two-chambered or three-chambered plastic unit with vertical columns bringing measurements marked in milliliters. The thoracic drainage devices cover a wide range and have evolved considerably since their introduction. The basic design principle of these systems has been the avoidance of air entrance in the pleural cavity during the various phases of the respiratory cycle and continuous drainage of air and fluid from the pleural cavity. A key issue in the successful treatment of patients is the understanding of how these systems function. The application and development was based on the original one-bottle system. The understanding of this principal system introduces us to the mechanism of function.
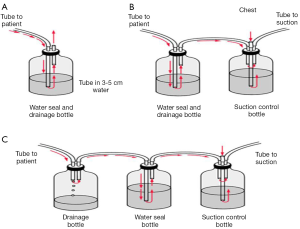
One-bottle system (Figure 1A)
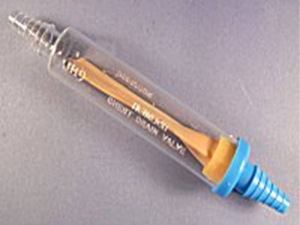
It consists of a bottle which collects and contains the fluid and at the same time seals air leak (leakage barrier-water seal). A rigid straw is immersed into the bottle, so that its tip is located 2 cm below the surface of the saline solution, which is put into the bottle. The other end of this rigid straw is connected to the thoracic drainage tube placed in the pleural cavity. In order to decompress the pressure from the air leak, there is an opening of one-way decompression valve (vent) through which the system is depressurized. It is important to remove this valve cover before connecting the system to the patient.
When the pleural pressure is positive, the pressure in the rigid straw becomes positive, and if the positive pressure in the rigid straw is greater than the depth to which the tube is immersed in the saline solution, then air will enter in the bottle and then depressurized by vent into the atmosphere. If the pleural pressure is negative, it will move liquid from the bottle to the rigid straw and air will not enter the pleural cavity or the rigid straw. This system is called water seal because the water bottle seals the pleural cavity from the air or liquid from the outside of the body. Just like a straw in a drink, air can push through the straw, but air can’t be drawn back up the straw.
It is clear that when the rigid straw is above the liquid level in the bottle, the system will not operate consistently developing pneumothorax.
However, when a significant quantity of liquid is drained from the pleural cavity of the patient, the liquid level will rise, thus requiring a greater pressure on the rigid straw to remove effectively additional air from the pleural cavity to the bottle. Practically, this system works if only air is leaving the chest, because if fluid is draining, it will add to the fluid in the water seal, and increase the depth, and as the depth increases, it becomes harder for the air to push through a higher level of water, and could result in air staying in the chest. As a result, the one-bottle system works efficiently for uncomplicated pneumothorax.
Another disadvantage of this system is that the positioning of the bottle at a level higher than the patient’s chest causes liquid passing into the pleural cavity (1).
Two-compartment system (Figure 1B)
For the aforementioned reasons of inefficient function of the one-bottle system in cases of pleural fluid effusion, it has been introduced the two-compartment system. This system is preferred over one-bottle system when large quantities of liquid are drained from the pleural cavity. With this system, the first bottle (closer to the patient) collects the drainage and the second bottle is the water seal, which remains at 2 cm (water seal and air vent). Therefore, the degree of water seal does not increase as fluid accumulates in the drain bottle. The water-seal bottle is the key for chest drainage, as it includes a place for drainage to collect and a one-way valve that prevents air or fluid from returning to the chest. Both the one and two-bottle chest drainage systems rely on gravity to create a pressure gradient by which air and fluid leave the chest. Keeping the drainage system below the level of the patient's chest enhances gravity drainage; additional pressure is created when the patient exhales or coughs. However, if the patient has a large air leak into the pleural space, gravity drainage may not be sufficient to evacuate the chest, and suction may be required. This also means the addition of a third bottle to the system—a suction control bottle.
Three-compartment system (Figure 1C)
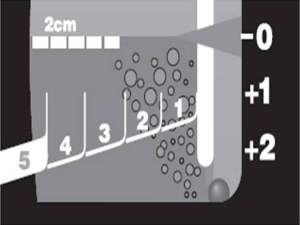
In 1967, Deknatel introduced the first integrated disposable chest drainage unit based on the three bottle system. The main rational of this approach was at this time that that suction was always required to pull air and fluid out of the pleural space and pull the lung up against the parietal pleura. If suction is required, a third bottle is added. However, recent research has shown that suction may actually prolong air leaks from the lung by pulling air through the opening that would otherwise close on its own (2,3). One of the chambers of the unit is the collection one. The patient tubing connects the drainage unit directly to the chest tube. Any drainage from the chest flows into this chamber. The collection chamber has to be calibrated and has a write-on surface to allow for easy measurement and recording of the time, date, and amount of drainage. The middle chamber of a traditional chest drainage system is the water seal. The main purpose of the water seal is to allow air to exit from the pleural space on exhalation and prevent air from entering the pleural cavity or mediastinum on inhalation. When the water seal chamber is filled with sterile fluid up to the 2 cm line, a 2 cm water seal is established. To maintain an effective seal, it is important to keep the chest drainage unit upright at all times and to monitor the water level in the water seal to check for evaporation. Sometimes it is necessary to apply negative pressure in the pleural cavity in order to facilitate re-expansion of the underlying lung parenchyma or accelerate air removal from the pleural cavity. The addition of a third bottle allows the controlled application of suction. A vent in the suction control bottle is connected to a vent on the water seal bottle. The two bottles are interconnected. The suction control bottle has a rigid straw similar to that of the water-seal bottle. The amount of negative pressure within the suction system is equal to the depth of immersion of the rigid straw below the liquid surface of the bottle. This size can be changed by adjusting the position of the rigid straw to the suction control bottle or by changing the depth of liquid in the bottle. The uncontrolled suction can have effects quite different to the expected results. Intensifying suction maximizes air leak, and creates more noise, annoyance, discomfort, favors the evaporation of liquid from the control bottle and, as it exacerbates air leak, prolongs hospital stay due to the tube maintenance with increased morbidity (1). Bubbling in the water seal chamber indicates an air leak. Air leak management remains the main problem in the discussion of the pneumothorax. Tools in the reliable diagnosis of the air leak are required to lead therapeutic decisions and further manipulations. Consensus on this item is hard enough; thus, objectivity is demanded and many efforts have been done to introduce and establish objective, reproducible classification system of the air leaks. The studies have shown that water seal is superior to wall suction to help stop most leaks. Even in patients with a pneumothorax and an air leak, water seal is safe and best; however, if a patient has a large leak (greater than an expiratory 3 on the classification system) or experiences subcutaneous emphysema or an expanding pneumothorax that causes hypoxia, then some suction (−10 cm of water) should be applied to the chest tubes (4-6). The patient air leak meter existing in some systems indicates the approximate degree of air leak from the chest cavity. The meter is made up of numbered columns, labeled from 1 (low) to 7 (high). The higher the numbered column through which bubbling occurs, the greater the degree of air leak (Figure 2). By documenting the number, the clinician can monitor air leak increase or decrease.
The water seal chamber may also have a calibrated manometer to measure the amount of negative pressure within the pleural cavity. The water level in the small arm of the water seal rises as intrapleural pressure becomes more negative. If there is no air leak, the water level should rise and fall with the patient’s respirations, reflecting normal pressure changes in the pleural cavity. During spontaneous respirations, the water level should rise during inhalation and fall during exhalation. If the patient is receiving positive pressure ventilation, the oscillation will be just the opposite the water level should fall with inhalation and rise with exhalation. This oscillation is called tidaling and is one indicator of a patent pleural chest tube. Some units have an anti-siphoning float valve in the water seal fluid column that prevents the water from being siphoned out of the water seal chamber and into the collection chamber during situations that create high negative pressures, such as chest tube stripping.
The original design of the float valve at the top of this chamber permitted uncontrolled vacuum levels to accumulate in the patient's chest with each subsequent stripping of the patient tube. To eliminate this pressure accumulation, manufacturers have also added manual high negative pressure relief valves to chest drain systems that allow filtered atmospheric air to enter the system to prevent any accumulation of negative pressure in the patient. However, with manual devices, the clinician must recognize the condition of high negativity, evidenced by the rise in the water level in the water seal, and depress the relief valve to remedy the situation.
Three situations can cause high negative pressure: (I) the patient in respiratory distress, coughing vigorously, or crying; (II) chest tube stripping; (III) decreasing or disconnecting suction. Vigorous milking or stripping can create dangerously high negative pressures. Research has documented negative pressures as high as −450 cm H2O. The system prevents accumulation of excessive high negative pressure as discussed above; however, the transient high negative pressures created by vigorous stripping can put the patient at risk for mediastinal trauma and graft trauma. We have to manipulate with caution and follow established hospital protocols. As it has been already mentioned, a manual high negativity relief valve is located on top of chest drainage systems. Depressing the high negativity relief valve allows filtered air into the system, relieving negativity and allowing the water level to return to baseline in the water seal. We have to use the high negativity relief valve with caution. If suction is not operative, or if operating on gravity drainage, depressing the high negativity relief valve can reduce negative pressure within the collection chamber to zero (atmosphere) with the resulting possibility of a pneumothorax (7).
Wet suction control
The chamber on the left side of the unit is the suction control chamber. Traditional chest drainage units regulate the amount of suction by the height of a column of water in the suction control chamber. A suction pressure of −20 cm H2O is commonly recommended. Lower levels may be indicated for infants and for patients with friable lung tissue, or if ordered by the physician. In a wet suction control system, the suction control chamber is filled to the desired height with sterile fluid and the short suction tubing is connected to a suction source, which is adjusted to produce gentle bubbling in the suction control chamber. Increasing suction at the suction source will increase airflow through the system, but will have minimal effect on the amount of suction imposed on the chest cavity. Excessive source suction not only causes loud bubbling (which can disturb patients and caregivers), but also hastens evaporation of water from the suction control chamber. These results in a lower amount of suction applied to the patient as the level of water decreases.
Dry suction
The next step in the evolution of chest drainage units was the development of dry suction control chambers. Dry suction control systems provide many advantages: higher suction pressure levels can be achieved, set-up is easy, and there is no fluid to evaporate which would decrease the amount of suction applied to the patient.
Instead of regulating the level of suction with a column of water, the dry suction units are controlled by a self-compensating regulator. To set the suction setting, the dial is rotated at the prescribed suction level. Suction can be set at −10, −15, −20, −30, or −40 cm of water. The unit is usually pre-set at −20 cm of water when opened. Connect the short suction tubing or suction port to the suction source. Source suction must be capable of delivering a minimum of 16 liters per minute (LPM) air flow. The setting of the suction control dial determines the approximate amount of suction imposed regardless of the amount of source suction. Patient situations that may require higher suction pressures of −30 or −40 cm H2O include: a large air leak from the lung surface, empyema or viscous pleural effusion, a reduction in pulmonary compliance, or anticipated difficulty in expansion of the pulmonary tissue to fill the hemithorax.
In the presence of a large air leak, air flow may be increased by increasing source suction, without increasing imposed negativity. It is not necessary to change the suction setting on the system to accommodate high air flows.
The suction control level can be changed at any time as prescribed by simply rotating the dial to the new suction setting (7). A drawback dry suction system is that it does not provide the same level of patient assessment information as a conventional water seal; for example, the clinician cannot see changes in the water level reflecting pressure changes in the chest. For optional air leak detection, a separate air leak monitor can be filled with water. A vacuum indicator on the face of the drain provides visual evidence of negative pressure (vacuum) inside the collection chamber (8).
To clamp or not to clamp
The decision whether to clamp a chest tube when the drainage system has been knocked over and disconnected or otherwise disrupted is based on your initial assessment of the water seal chamber and air leak meter. If there has been no bubbling in the water seal, you can deduce there is no air leak from the lung. Therefore, the tube may be clamped for the short time it takes to reestablish drainage. If there has been bubbling and your assessment has determined there is an air leak from the lung, you must not clamp the chest tube. Doing so will cause air to accumulate in the pleural cavity since the air has no means of escape. This can rapidly lead to tension pneumothorax. The few times you should clamp a chest tube are when: (I) you are performing a physician-ordered procedure such as sclerosing; (II) assessing for a leak or; (III) prior to removing the chest tube to determine if the patient can do without the chest tube (with a physician order). You should never clamp a chest tube during patient transport unless the chest drainage system becomes disrupted during patient movement, and then only if there is no air leak (7,9-11).
What we have to take care of
- Keep the system closed and below chest level. Make sure all connections are taped and the chest tube is secured to the chest wall;
- Ensure that the suction control chamber is filled with sterile water to the 20 cm-level or as prescribed. If using suction, make sure the suction unit’s pressure level causes slow but steady bubbling in the suction control chamber;
- Make sure the water-seal chamber is filled with sterile water to the level specified by the manufacturer. You should see fluctuation (tidaling) of the fluid level in the water-seal chamber; if you don’t, the system may not be patent or working properly, or the patient’s lung may have reexpanded;
- Look for constant or intermittent bubbling in the water-seal chamber, which indicates leaks in the drainage system. Identify and correct external leaks. Notify the health care provider immediately if you can’t identify an external leak or correct it;
- Assess the amount, color, and consistency of drainage in the drainage tubing and in the collection chamber. Mark the drainage level on the outside of the collection chamber (with date, time, and initials) every 8 hours or more frequently if indicated. Report drainage that’s excessive, cloudy, or unexpectedly bloody;
- Encourage the patient to perform deep breathing, coughing, and incentive spirometry. Assist with repositioning or ambulation as ordered. Provide adequate analgesia;
- Assess vital signs, breath sounds, SpO2, and insertion site for subcutaneous emphysema as ordered;
- When the chest tube is removed, immediately apply sterile occlusive petroleum gauze dressing over the site to prevent air from entering the pleural space;
- Don’t let the drainage tubing kink, loop, or interfere with the patient’s movement;
- Don’t clamp a chest tube, except momentarily when replacing the chest drainage unit, assessing for an air leak, or assessing the patient’s tolerance of chest tube removal, and during chest tube removal;
- Don’t aggressively manipulate the chest tube; don’t strip or milk it;
- A patient who is free from pain, to the degree that an effective cough can be produced, will generate a much higher pressure than can safely be produced with suction;
- If a patient cannot re-inflate his own lung, high volume, low pressure "thoracic" suction in the range of 15-25 cm of water can help;
- Patients on mechanical ventilators cannot produce an effective cough and therefore suction is advised;
- Close surveillance is required by nursing staff trained to recognize faults in the drainage and suction system. It is better to remove suction than to use a faulty device;
- The depth of the water in the suction bottle determines the amount of negative pressure that can be transmitted to the chest, NOT the reading on the vacuum regulator;
- There is no research to support this number of −20 cm H2O, just convention. Higher negative pressure can increase the flow rate out of the chest, but it can also damage tissue;
- The water seal chamber and suction control chamber provide intrathoracic pressure monitoring. Remember that in gravity drainage without suction the level of water in the water seal chamber = intrathoracic pressure;
- Slow, gradual rise in water level over time means more negative pressure in pleural space and signals healing. Goal is to return to −8 cm H2O;
- When we apply suction: Level of water in suction control + level of water in water seal chamber = intrathoracic pressure.
Heimlich valve (Figure 3)
This valve (also known as the Heimlich valve after its inventor, Henry Heimlich) is a rubber flutter one-way valve within a rigid plastic tube which connects to standard chest drain. It does not need to be kept upright like the underwater sealed drain and therefore is suitable for outpatient use. However, the efferent portal of the Heimlich valve must be kept open to the atmosphere making control of the fluid effluent difficult. The device is bulky under clothing and staining is a constant problem. To avoid this problem, the valve should be attached to a perforated plastic bag, or a specifically designed one-way valve including a small reservoir can be used. Other units for outpatient management incorporating a system for checking air leaks are available, but are more expensive than a classic Heimlich or a plastic bag (12).
The use of this one-way valve has been proposed in the outpatient management of first episodes of primary spontaneous pneumothorax (13,14), in the early postoperative discharge after lobectomy or segmentectomy for lung cancer in fast-tracking protocols (15-17), in complicated postoperative air leaks etc(18).
Technological advancement: digital systems (Figure 4)
Despite the dramatic advance that the air leak meter has provided in the management of patients with leaks, the presence or absence of a leak still has subjective overtones and has not been completely quantified. For example, it is not uncommon for physicians to ask one another as they stare at an air leak chamber, “Was there really a bubble on that breath, was there really a leak, or was it just a momentum leak and the clearing of air with the first cough?” Often one experienced observer at the bedside will report a leak while another disagrees. Given the trend in the third millennium towards digitalization in many facets of life and especially in science, it is no surprise that several companies have applied digital technology to objectively measure the size of an air leak out a chest tube (19). There are currently systems which generate the flows by digital meters incorporated in the drainage portals. Advanced chest drainage digital systems allow early mobilization of the patients with suction and accurate charting of air leak. They have scientific digital flow recordings with an in built alarm system. There are various alarms which alert the nurses regarding blocks, high volumes and battery status. The device also flushes the collection tubing connected to the inter-costal drain preventing blockage of drains.
The clinicians can assess air-leak in a scientific and objective manner as the data can be reviewed in a graphic format. Drain removal is performed when there is minimal flow and the graphs are stable. It has been reported in a randomized trial that the use of such an electronic chest drainage device was associated with a cost saving of approximately €500 per patient (20). For another one it has been demonstrated two days’ shorter duration of chest tube usage, and a 1.5-day shorter hospital stay, with a consequent saving of approximately €750 per patient (21). An ideal digital chest drainage system has the following characteristics: large friendly reservoir for fluid collection and analysis; functional in different levels of suction; compact to permit patient ambulation; latex-free, quiet, tip-over safe, reusable, and inexpensive; digital continuous accurate measurement of the amount of the chest tube drainage and the size of air leaks; written record of events in the pleural space; easy for both staff and patients to use; allows for the patient to be sent home on the same device; data available to the nurses’ station or physician’s office for assessment (22-39). There is no question that the future belongs to new technologies. However, it depends on many parameters the introduction of digital devices: the quality superiority, the familiarization of the medical and nursing staff, the education and the training, the culture of technological advancement adoption, the availability of sources, and the cost-effectiveness are some of them (40-59). For the specialists, most efficient device is usually the most familiar to them, but air leak remains a medical challenge and sometimes needs sophisticated approach and flexibility to give reliable solution, secure quality of life, no pain or discomfort and at the same time reduce the cost (50,60-66).
Acknowledgements
Disclosure: The authors declare no conflict of interest.
References
- Light RW. eds. Pleural diseases. 3rd Edition. Williams & Wilkins, 1995:330-2.
- Coughlin SM, Emmerton-Coughlin HM, Malthaner R. Management of chest tubes after pulmonary resection: a systematic review and meta-analysis. Can J Surg 2012;55:264-70. [PubMed]
- Cerfolio RJ. Recent advances in the treatment of air leaks. Curr Opin Pulm Med 2005;11:319-23. [PubMed]
- Cerfolio RJ, Bass C, Katholi CR. Prospective randomized trial compares suction versus water seal for air leaks. Ann Thorac Surg 2001;71:1613-7. [PubMed]
- Cerfolio RJ, Bryant AS, Singh S, et al. The management of chest tubes in patients with a pneumothorax and an air leak after pulmonary resection. Chest 2005;128:816-20. [PubMed]
- Marshall MB, Deeb ME, Bleier JI, et al. Suction vs water seal after pulmonary resection: a randomized prospective study. Chest 2002;121:831-5. [PubMed]
- Chest Drainage Systems. Available online: http://www.teleflex.com/en/usa/ucd/chest_drainage_systems.php
- Gillespie BM, Rickard CM, Thalib L, et al. Use of Negative-Pressure Wound Dressings to Prevent Surgical Site Complications After Primary Hip Arthroplasty: A Pilot RCT. Surg Innov 2015. [Epub ahead of print].
- Shuster PM. Chest tubes: to clamp or not to clamp. Nurse Educ 1998;23:9,13.
- Gupta N. Pneumothorax: is chest tube clamp necessary before removal? Chest 2001;119:1292-3. [PubMed]
- Williams T. To clamp or not to clamp? Nurs Times 1992;88:33. [PubMed]
- Varela G, Jiménez MF, Novoa N. Portable chest drainage systems and outpatient chest tube management. Thorac Surg Clin 2010;20:421-6. [PubMed]
- Massongo M, Leroy S, Scherpereel A, et al. Outpatient management of primary spontaneous pneumothorax: a prospective study. Eur Respir J 2014;43:582-90. [PubMed]
- Brims FJ, Maskell NA. Ambulatory treatment in the management of pneumothorax: a systematic review of the literature. Thorax 2013;68:664-9. [PubMed]
- McKenna RJ Jr, Mahtabifard A, Pickens A, et al. Fast-tracking after video-assisted thoracoscopic surgery lobectomy, segmentectomy, and pneumonectomy. Ann Thorac Surg 2007;84:1663-7; discussion 1667-8.
- Rice TW, Okereke IC, Blackstone EH. Persistent air-leak following pulmonary resection. Chest Surg Clin N Am 2002;12:529-39. [PubMed]
- Cerfolio RJ, Bass CS, Pask AH, et al. Predictors and treatment of persistent air leaks. Ann Thorac Surg 2002;73:1727-30; discussion 1730-1.
- Rathinam S, Steyn RS. Management of complicated postoperative air-leak - a new indication for the Asherman chest seal. Interact Cardiovasc Thorac Surg 2007;6:691-4. [PubMed]
- Certfolio RJ. Clinical use of a digital air leak system. Available online: http://www.ctsnet.org/portals/thoracic/newtechnology/article-13
- Brunelli A, Salati M, Refai M, et al. Evaluation of a new chest tube removal protocol using digital air leak monitoring after lobectomy: a prospective randomised trial. Eur J Cardiothorac Surg 2010;37:56-60. [PubMed]
- Pompili C, Brunelli A, Salati M, et al. Impact of the learning curve in the use of a novel electronic chest drainage system after pulmonary lobectomy: a case-matched analysis on the duration of chest tube usage. Interact Cardiovasc Thorac Surg 2011;13:490-3; discussion 493. [PubMed]
- Cerfolio RJ, Varela G, Brunelli A. Digital and smart chest drainage systems to monitor air leaks: the birth of a new era? Thorac Surg Clin 2010;20:413-20. [PubMed]
- Kioumis IP, Zarogoulidis K, Huang H, et al. Pneumothorax in cystic fibrosis. J Thorac Dis 2014;6:S480-7. [PubMed]
- Kuhajda I, Zarogoulidis K, Kougioumtzi I, et al. Tube thoracostomy; chest tube implantation and follow up. J Thorac Dis 2014;6:S470-9. [PubMed]
- Manika K, Kioumis I, Zarogoulidis K, et al. Pneumothorax in sarcoidosis. J Thorac Dis 2014;6:S466-9. [PubMed]
- Kuhajda I, Zarogoulidis K, Kougioumtzi I, et al. Penetrating trauma. J Thorac Dis 2014;6:S461-5. [PubMed]
- Visouli AN, Zarogoulidis K, Kougioumtzi I, et al. Catamenial pneumothorax. J Thorac Dis 2014;6:S448-60. [PubMed]
- Huang Y, Huang H, Li Q, et al. Transbronchial lung biopsy and pneumothorax. J Thorac Dis 2014;6:S443-7. [PubMed]
- Terzi E, Zarogoulidis K, Kougioumtzi I, et al. Acute respiratory distress syndrome and pneumothorax. J Thorac Dis 2014;6:S435-42. [PubMed]
- Boskovic T, Stojanovic M, Stanic J, et al. Pneumothorax after transbronchial needle biopsy. J Thorac Dis 2014;6:S427-34. [PubMed]
- Li Z, Huang H, Li Q, et al. Pneumothorax: observation. J Thorac Dis 2014;6:S421-6. [PubMed]
- Huang Y, Huang H, Li Q, et al. Approach of the treatment for pneumothorax. J Thorac Dis 2014;6:S416-20. [PubMed]
- Browning RF, Parrish S, Sarkar S, et al. Bronchoscopic interventions for severe COPD. J Thorac Dis 2014;6:S407-15. [PubMed]
- Machairiotis N, Kougioumtzi I, Dryllis G, et al. Laparoscopy induced pneumothorax. J Thorac Dis 2014;6:S404-6. [PubMed]
- Ouellette DR, Parrish S, Browning RF, et al. Unusual causes of pneumothorax. J Thorac Dis 2014;6:S392-403. [PubMed]
- Parrish S, Browning RF, Turner JF Jr, et al. The role for medical thoracoscopy in pneumothorax. J Thorac Dis 2014;6:S383-91. [PubMed]
- Terzi E, Zarogoulidis K, Kougioumtzi I, et al. Human immunodeficiency virus infection and pneumothorax. J Thorac Dis 2014;6:S377-82. [PubMed]
- Zarogoulidis P, Kioumis I, Pitsiou G, et al. Pneumothorax: from definition to diagnosis and treatment. J Thorac Dis 2014;6:S372-6. [PubMed]
- Tsakiridis K, Mpakas A, Kesisis G, et al. Lung inflammatory response syndrome after cardiac-operations and treatment of lornoxicam. J Thorac Dis 2014;6 Suppl 1:S78-98. [PubMed]
- Tsakiridis K, Zarogoulidis P, Vretzkakis G, et al. Effect of lornoxicam in lung inflammatory response syndrome after operations for cardiac surgery with cardiopulmonary bypass. J Thorac Dis 2014;6 Suppl 1:S7-20. [PubMed]
- Argiriou M, Kolokotron SM, Sakellaridis T, et al. Right heart failure post left ventricular assist device implantation. J Thorac Dis 2014;6 Suppl 1:S52-9. [PubMed]
- Madesis A, Tsakiridis K, Zarogoulidis P, et al. Review of mitral valve insufficiency: repair or replacement. J Thorac Dis 2014;6 Suppl 1:S39-51. [PubMed]
- Siminelakis S, Kakourou A, Batistatou A, et al. Thirteen years follow-up of heart myxoma operated patients: what is the appropriate surgical technique? J Thorac Dis 2014;6 Suppl 1:S32-8. [PubMed]
- Foroulis CN, Kleontas A, Karatzopoulos A, et al. Early reoperation performed for the management of complications in patients undergoing general thoracic surgical procedures. J Thorac Dis 2014;6 Suppl 1:S21-31. [PubMed]
- Nikolaos P, Vasilios L, Efstratios K, et al. Therapeutic modalities for Pancoast tumors. J Thorac Dis 2014;6 Suppl 1:S180-93. [PubMed]
- Koutentakis M, Siminelakis S, Korantzopoulos P, et al. Surgical management of cardiac implantable electronic device infections. J Thorac Dis 2014;6 Suppl 1:S173-9. [PubMed]
- Spyratos D, Zarogoulidis P, Porpodis K, et al. Preoperative evaluation for lung cancer resection. J Thorac Dis 2014;6 Suppl 1:S162-6. [PubMed]
- Porpodis K, Zarogoulidis P, Spyratos D, et al. Pneumothorax and asthma. J Thorac Dis 2014;6 Suppl 1:S152-61. [PubMed]
- Panagopoulos N, Leivaditis V, Koletsis E, et al. Pancoast tumors: characteristics and preoperative assessment. J Thorac Dis 2014;6 Suppl 1:S108-15. [PubMed]
- Visouli AN, Darwiche K, Mpakas A, et al. Catamenial pneumothorax: a rare entity? Report of 5 cases and review of the literature. J Thorac Dis 2012;4 Suppl 1:17-31. [PubMed]
- Zarogoulidis P, Chatzaki E, Hohenforst-Schmidt W, et al. Management of malignant pleural effusion by suicide gene therapy in advanced stage lung cancer: a case series and literature review. Cancer Gene Ther 2012;19:593-600. [PubMed]
- Papaioannou M, Pitsiou G, Manika K, et al. COPD assessment test: a simple tool to evaluate disease severity and response to treatment. COPD 2014;11:489-95. [PubMed]
- Boskovic T, Stanic J, Pena-Karan S, et al. Pneumothorax after transthoracic needle biopsy of lung lesions under CT guidance. J Thorac Dis 2014;6 Suppl 1:S99-107. [PubMed]
- Papaiwannou A, Zarogoulidis P, Porpodis K, et al. Asthma-chronic obstructive pulmonary disease overlap syndrome (ACOS): current literature review. J Thorac Dis 2014;6 Suppl 1:S146-51. [PubMed]
- Zarogoulidis P, Porpodis K, Kioumis I, et al. Experimentation with inhaled bronchodilators and corticosteroids. Int J Pharm 2014;461:411-8. [PubMed]
- Bai C, Huang H, Yao X, et al. Application of flexible bronchoscopy in inhalation lung injury. Diagn Pathol 2013;8:174. [PubMed]
- Zarogoulidis P, Kioumis I, Porpodis K, et al. Clinical experimentation with aerosol antibiotics: current and future methods of administration. Drug Des Devel Ther 2013;7:1115-34. [PubMed]
- Zarogoulidis P, Pataka A, Terzi E, et al. Intensive care unit and lung cancer: when should we intubate? J Thorac Dis 2013;5 Suppl 4:S407-12. [PubMed]
- Hohenforst-Schmidt W, Petermann A, Visouli A, et al. Successful application of extracorporeal membrane oxygenation due to pulmonary hemorrhage secondary to granulomatosis with polyangiitis. Drug Des Devel Ther 2013;7:627-33. [PubMed]
- Zarogoulidis P, Kontakiotis T, Tsakiridis K, et al. Difficult airway and difficult intubation in postintubation tracheal stenosis: a case report and literature review. Ther Clin Risk Manag 2012;8:279-86. [PubMed]
- Zarogoulidis P, Tsakiridis K, Kioumis I, et al. Cardiothoracic diseases: basic treatment. J Thorac Dis 2014;6 Suppl 1:S1. [PubMed]
- Kolettas A, Grosomanidis V, Kolettas V, et al. Influence of apnoeic oxygenation in respiratory and circulatory system under general anaesthesia. J Thorac Dis 2014;6 Suppl 1:S116-45. [PubMed]
- Turner JF, Quan W, Zarogoulidis P, et al. A case of pulmonary infiltrates in a patient with colon carcinoma. Case Rep Oncol 2014;7:39-42. [PubMed]
- Machairiotis N, Stylianaki A, Dryllis G, et al. Extrapelvic endometriosis: a rare entity or an under diagnosed condition? Diagn Pathol 2013;8:194. [PubMed]
- Tsakiridis K, Zarogoulidis P. An interview between a pulmonologist and a thoracic surgeon-Pleuroscopy: the reappearance of an old definition. J Thorac Dis 2013;5 Suppl 4:S449-51. [PubMed]
- Huang H, Li C, Zarogoulidis P, et al. Endometriosis of the lung: report of a case and literature review. Eur J Med Res 2013;18:13. [PubMed]