
Estimated glomerular filtration rate, anemia and outcomes in patients with ischemic stroke
Introduction
Chronic kidney disease (CKD) is a common independent risk factor and comorbidity for stroke (1,2). In most patients with CKD, anemia is highly prevalent and is known as an intermediate marker of CKD; furthermore, anemia is regarded as a strong independent predictor of increased mortality after stroke (3). A previous study on the community-based population demonstrated that CKD combined with anemia was significantly associated with an increased risk of stroke (4). Moreover, in stroke patients, the presence of CKD and anemia predicted the high risk of mortality at 1-year of follow-up (5). However, this retrospective study only included first-ever diagnosed stroke patients and did not analyze other meaningful outcomes besides mortality. In addition, the combined relationship of CKD and anemia with stroke outcome has not been well studied.
We thereby hypothesized that patients with modest or severe kidney dysfunction [estimated glomerular filtration rate (eGFR) <60 mL/min per 1.73 m2] and anemia had a strong association with poorer clinical outcomes compared to those without kidney dysfunction or anemia. We tested this assumption in the prospective cohort study of China National Stroke Registry (CNSR).
Methods
Study population
All ischemic stroke patients enrolled in CNSR between 2007 and 2008 were eligible to enter this analysis, excluding those potentially cause of anemia, such as immune deficiency, pregnancy, cancer, gastrointestinal bleeding (GI bleeding), end-stage of liver or extremely low body mass index (BMI) ≤18.5. The rationale and design of CNSR has been described previously (6). Briefly, CNSR was a prospective, multicenter, cohort study that enrolled 12,415 patients with acute cerebrovascular events from 132 hospitals across China. The study was approved by the ethics committee of Beijing Tiantan Hospital. Informed consents were obtained from all patients who were enrolled in the CNSR.
Data collection
Trained interviewers (neurologists from the included hospitals) conducted the data collection. Baseline information included age and sex. Vascular risk factors included hypertension, diabetes mellitus, hyperlipidemia, atrial fibrillation, coronary heart disease, congestive heart failure, stroke history, family stroke history, current smoking, and heavy drinking. Clinical features included the National Institutes of Health Stroke Scale (NIHSS) on admission, serum levels of creatinine, hemoglobin and fasting blood glucose. Stroke subtypes were classified by TOAST criteria (Trial of ORG 10172 in acute stroke treatment) (7). The complications during hospitalization included pneumonia and urinary tract infection (UTI). In addition, in-hospital medications included antiplatelet, anticoagulation, lipid-lowering agents, hypertensive agents, and hypoglycemic agents. Heavy drinking was defined as ≥2 standardized alcohol drinks every day.
Renal function and anemia assessment
The eGFR was calculated by using the Chronic Kidney Disease Epidemiology Collaboration creatinine equation (CKD-EPI) with an adjusted coefficient of 1.1 for the Asian population (8,9). The serum creatinine level was measured on admission by using the Jaffe method and was reduced by 5% for the Chronic Kidney Disease Epidemiology Collaboration equation (10). The eGFR level was dichotomized into low eGFR (less than 60 mL/min per 1.73 m2) and normal eGFR (more than 60 mL/min per 1.73 m2), a cutting-off point that has been previously proposed to distinguish moderate or severe CKD from fine renal function (11). Anemia was defined as a hemoglobin value of less than 120 g/L in women and 130 g/L in men according to the World Health Organization (WHO) criteria (12). According to the eGFR and hemoglobin levels, patients were categorized into four groups: low eGFR with or without anemia, and normal eGFR with or without anemia.
Follow-up and outcome estimation
Trained interviewers based on a unified protocol performed a telephone follow-up at 3 months and 1 year after stroke onset. The outcome was measured by poor functional outcome, all-cause mortality and stroke recurrence. The poor functional outcome was defined as a score of 3 to 6 on the modified Rankin Scale (mRS) (13). Death was confirmed by the death certificate from either the local citizen registry or the hospital where the patient was treated. Stroke recurrence was defined as a worsening of the index neurological deficit (NIHSS increasing ≥4, excluding hemorrhagic transformation after infarction or symptomatic intracranial hemorrhage), a new neurological deficit, or re-hospitalization with a diagnosis of ischemic or hemorrhagic stroke (13).
Statistics
The baseline and clinical characteristics were compared between these four groups by using the χ2 test or Fisher exact test for categorical variables and one-way ANOVA or Kruskal-Wallis test for continuous variables with a normal or skewed distribution respectively. Continuous variables were expressed as means with standard deviation (SD) or medians with interquartile range (IQR), as appropriate. Categorical data was presented as proportions.
Univariate and multivariate logistic regression models were used to evaluate the association between the clinical outcome and eGFR with or without anemia. All significant covariates in the univariate regression analysis were included in the multivariate model. Odds ratios (ORs) with 95% confidence intervals (CIs) were calculated by using those with normal eGFR and without anemia as reference. To examine the interaction effect between eGFR and hemoglobin, we tested the statistical significance of eGFR × hemoglobin in a multivariate logistical regression model.
All analyses were conducted with SAS version 9.4 software (SAS Institute Inc., Cary, NC, USA). Two-tailed P values <0.05 were considered to be statistically significant.
Results
Subjects recruited
In this study, 12,415 ischemic stroke patients were enrolled, 900 of whom had immune deficiency, pregnancy, cancer, GI bleeding, end-stage of liver or extremely low BMI ≤18.5 and therefore were excluded. Another 3,197 (25.8%) individuals without data on serum creatinine (n=3,050) or hemoglobin (n=2,514) were also excluded. Fifteen patients were excluded for no follow-up data. A total of 8,303 patients were included in the final analysis (Figure 1).
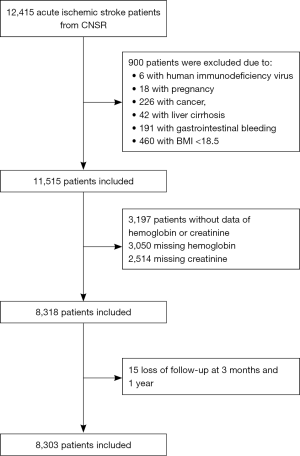
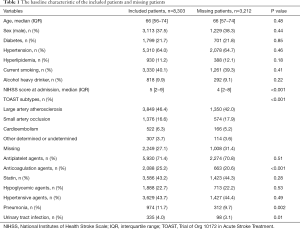
There was no significant disparity within the demography and common vascular risk factors between included and missing patients. No significance was detected in the treatment for hypertension, diabetes mellitus, use of antiplatelet agents, and statin, except those missing were less likely to use anticoagulation agents possibly because of bleeding diathesis. In addition, the included patients had a higher baseline NIHSS score and stroke etiology was imbalanced between the two groups yet (Table 1).
Full table
Baseline characteristics
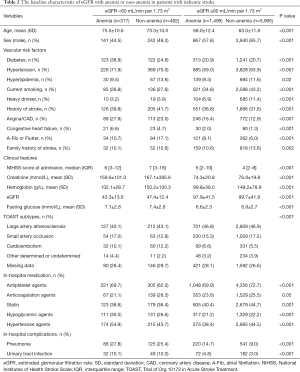
The baseline and clinical features of the included patients are demonstrated in Table 2. Overall, the subjects with both low eGFR and anemia accounted for 3.8% of the total studied population and 39.2% of the low eGFR population. These subjects were more likely to be elderly and female. They were also more likely to have vascular risk factors including hypertension, diabetes mellitus, coronary artery disease, atrial fibrillation, heart failure, but were less likely to be currently smoking and heavily drinking. They received more antihypertensive and hypoglycemic medications. However, the subjects with normal eGFR and hemoglobin levels were more likely to receive antiplatelet and statin therapy. In-hospital pneumonia and UTI were more prevalent in the subjects with low eGFR and anemia.
Full table
Association of eGFR and anemia with clinical outcome at 3 months
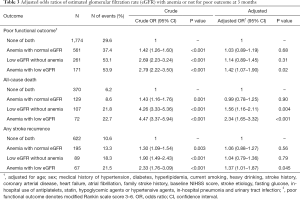
The association of eGFR and anemia with clinical outcomes after stroke at 3 months is presented in Table 3. In the univariate analysis, presence of low eGFR and/or anemia were associated with all of the adverse stroke outcomes (P<0.001). After adjusting for age, sex, medical history, the etiology of stroke, and other potential confounding factors, patients with low eGFR and normal hemoglobin had an increased risk of mortality [adjusted OR (aOR) =1.56; 95% CI, 1.16–2.11]. On the contrary, patients with anemia and normal eGFR were not associated with any 3-month poor outcome in this study. While patients with both low eGFR and anemia had increased risks of 3-month poor outcomes after the index stroke: poor functional outcome with aOR =1.42; 95% CI, 1.07–1.90; all-cause mortality with aOR =2.34; 95% CI, 1.65–3.32; any stroke recurrence with aOR =1.37; 95% CI, 1.01–1.87.
Full table
Association of eGFR and anemia with clinical outcome at 1 year
The association of eGFR and anemia with clinical outcomes at 1 year is shown in Table 4. Similarly, in the univariate analysis, patients with low eGFR or/and anemia were associated with poor functional outcome, all-cause mortality, and any stroke recurrence (all of these results, P<0.001), compared to those with normal eGFR and hemoglobin. After adjusting for age, sex and other potential confounding factors, low eGFR with normal hemoglobin merely increased the risk of mortality (aOR =1.50; 95% CI, 1.14–1.97, P=0.004). There was no significant association between anemia with normal eGFR and any poor outcome. Patients with low eGFR and anemia were associated with poor functional outcome (aOR =1.73; 95% CI, 1.30–2.29, P=0.001), all-cause mortality (aOR =2.64; 95% CI, 1.94–3.59, P<0.001), and any stroke recurrence (aOR =1.42; 95% CI, 1.06–1.91, P=0.02). Combined and interactive effects of the pattern of low eGFR and presence of anemia on the poor functional outcome (P for interaction =0.02) and all-cause mortality (P for interaction =0.046) were observed (Figure 2).
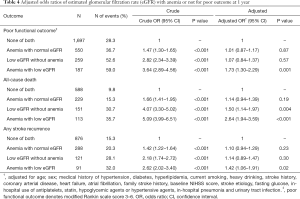
Full table

Discussion
In this Chinese national registry cohort study, patients with low eGFR and anemia were associated with an increased risk of poor functional outcome, all-cause mortality and any stroke recurrence after the index stroke at 3 months and 1 year. Furthermore, patients with low eGFR but normal hemoglobin had increased risks of all-cause mortality at 3 months and 1 year. However, patients with anemia but normal eGFR were not linked to any of the short- or long-term adverse endpoints after stroke.
Anemia is a common complication of CKD potentially due to the deficient erythropoietin (EPO) production (14).
Previous studies draw the inconsistent conclusions on the association between anemia and stroke outcomes (3,5,15-18). A cohort study showed anemia was an independent risk factor for the increased odds of 1-year mortality after the first-ever stroke (5). However, it included both ischemic and hemorrhagic stroke, and lacked specific analysis on the association between anemia and the risk of mortality in ischemic stroke patients. Another study focused on recanalized stroke patients and showed that baseline anemia was linked to an increased risk of poor outcome and mortality (15), in which only highly selected stroke patients treated with thrombolysis or endovascular therapy were included and without consideration of the status of the kidney function. Some studies had contrasting conclusions that failed to identify the association between anemia and unfavorable outcome after stroke (16-18). Sharma et al. documented that neither baseline anemia nor anemia during hospitalization was related with unfavorable functional outcome (16). As a retrospective study, it also did not consider the kidney status and other clinical outcomes. In our study, only anemia in the lower levels of eGFR was associated with poor outcome of acute ischemic stroke, while most of the previous studies did not offer such stratification. Our study indicates that the kidney function might play a role in the relationship of anemia and stroke outcome. It is worth mentioning that anemia was not further stratified based on the levels of hemoglobin in this study, which might underestimate the association between severe anemia in normal eGFR group and poor outcome after ischemic stroke.
The mechanisms of the association between low eGFR and anemia with poor outcome after stroke is insufficiently understood. For patients with chronically low eGFR, they would have worsening of water and sodium retention, development of left ventricular hypertrophy, the exacerbation of heart failure and accelerated atherosclerosis. The reduced EPO supposed to have a neuronal protective effect also might aggravate the poor outcomes after stroke (19-21). Furthermore, the presence of both low eGFR and anemia may potentially be an indication of severe CKD, which may have an adverse effect on stroke recovery.
To our knowledge, this is the largest study to date examining eGFR and anemia post stroke. When eGFR declined, the prevalence of anemia would increase, especially when eGFR was below 60 mL/min/1.73 m2. The presence of anemia was 1% to 9% at eGFR of 30 mL/min/1.73 m2 and was about 67% at eGFR of 15 mL/min/1.73 m2 (22). In our study, the incidence of anemia was increased to about 40% of stroke patients with low eGFR after excluding other potential etiological factors, such as cancer, GI bleeding, and liver cirrhosis, etc. In China, the burden of anemia associated with CKD may be substantial, given the large number of population with CKD (23). Our observation suggested that stroke patients with comorbidities of low eGFR and anemia would not have an optimistic outcome. EPO may be an option to these patients to prevent or improve their neurological function (24). However, EPO could increase the risk of thromboembolic events in patients with chronic heart failure without significant improvement in mortality or hospitalization (25). Therefore, more large studies are needed to further investigate the efficacy and safety of EPO treatment in CKD patients concomitant with anemia after stroke onset.
Study limitations
Our study has several limitations. First, our study did not stratify the severity and the exact causes of anemia. Data on the work up of anemia was not complete in the dataset. Severe anemia could affect the outcome of stroke. Second, it was difficult to determine the exact causes of low eGFR, from either CKD or acute kidney dysfunction. Third, our study focused on only Chinese adult patients with stroke and these results may not be generalizable to patients of other ethnicities. Lastly, other confounding factors, such as inflammation or undiagnosed malignancy, could cause both low eGFR and anemia and increase the risk of poor outcome.
Conclusions
The combination of baseline low eGFR and anemia was a stronger predictor to short- and long-term poor functional outcome, all-cause mortality and stroke recurrence in patients with acute ischemic stroke. Based on this finding, treatment of kidney insufficiency and correction of anemia earlier may potentially help to improve the outcome of patients with ischemic stroke
Acknowledgments
Funding: This study was supported by grants from the Ministry of Science and Technology of the People’s Republic of China (2016YFC0901002, 2016YFC0901001, 2017YFC1310901, 2017YFC1310902, 2017YFC1307905, 2018YFC1311700 and 2018YFC1311706), National Natural Science Foundation of China (81701149, 81971091), and Beijing Municipal Administration of Hospitals (SML20150502, QML20190501), grants from Jiangsu Provincial Medical Key Discipline Project (ZDXKB 2016022) and Suzhou Clinical Research Center of Neurological Disease (Szzx201503). This was also partly supported by the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD).
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. This study was approved by the ethics committee of Beijing Tiantan Hospital. Informed consents were obtained from all patients who were enrolled in CNSR.
References
- Luo Y, Wang X, Matsushita K, et al. Associations between estimated glomerular filtration rate and stroke outcomes in diabetic versus nondiabetic patients. Stroke 2014;45:2887-93. [Crossref] [PubMed]
- Yahalom G, Schwartz R, Schwammenthal Y, et al. Chronic kidney disease and clinical outcome in patients with acute stroke. Stroke 2009;40:1296-303. [Crossref] [PubMed]
- Barlas RS, Honney K, Loke YK, et al. Impact of Hemoglobin Levels and Anemia on Mortality in Acute Stroke: Analysis of UK Regional Registry Data, Systematic Review, and Meta-Analysis. J Am Heart Assoc 2016. [Crossref] [PubMed]
- Abramson JL, Jurkovitz CT, Vaccarino V, et al. Chronic kidney disease, anemia, and incident stroke in a middle-aged, community-based population: the ARIC Study. Kidney Int 2003;64:610-5. [Crossref] [PubMed]
- Del Fabbro P, Luthi JC, Carrera E, et al. Anemia and chronic kidney disease are potential risk factors for mortality in stroke patients: a historic cohort study. BMC Nephrol 2010;11:27. [Crossref] [PubMed]
- Wang Y, Cui L, Ji X, et al. The China National Stroke Registry for patients with acute cerebrovascular events: design, rationale, and baseline patient characteristics. Int J Stroke 2011;6:355-61. [Crossref] [PubMed]
- Adams HP Jr, Bendixen BH, Kappelle LJ, et al. Classification of subtype of acute ischemic stroke. Definitions for use in a multicenter clinical trial. TOAST. Trial of Org 10172 in Acute Stroke Treatment. Stroke 1993;24:35-41. [Crossref] [PubMed]
- Teo BW, Xu H, Wang D, et al. GFR estimating equations in a multiethnic Asian population. Am J Kidney Dis 2011;58:56-63. [Crossref] [PubMed]
- Wang X, Luo Y, Wang Y, et al. Comparison of associations of outcomes after stroke with estimated GFR using Chinese modifications of the MDRD study and CKD-EPI creatinine equations: results from the China National Stroke Registry. Am J Kidney Dis 2014;63:59-67. [Crossref] [PubMed]
- Skali H, Uno H, Levey AS, et al. Prognostic assessment of estimated glomerular filtration rate by the new Chronic Kidney Disease Epidemiology Collaboration equation in comparison with the Modification of Diet in Renal Disease Study equation. Am Heart J 2011;162:548-54. [Crossref] [PubMed]
- National Kidney F. K/DOQI clinical practice guidelines for chronic kidney disease: evaluation, classification, and stratification. Am J Kidney Dis 2002;39:S1-266. [PubMed]
- Nutritional anaemias. Report of a WHO group of experts. World Health Organ Tech Rep Ser 1972;503:1-29. [PubMed]
- Pan Y, Jing J, Chen W, et al. Post-Glucose Load Measures of Insulin Resistance and Prognosis of Nondiabetic Patients With Ischemic Stroke. J Am Heart Assoc 2017. [Crossref] [PubMed]
- Erslev AJ, Besarab A. Erythropoietin in the pathogenesis and treatment of the anemia of chronic renal failure. Kidney Int 1997;51:622-30. [Crossref] [PubMed]
- Bellwald S, Balasubramaniam R, Nagler M, et al. Association of anemia and hemoglobin decrease during acute stroke treatment with infarct growth and clinical outcome. PLoS One 2018;13:e0203535. [Crossref] [PubMed]
- Sharma K, Johnson DJ, Johnson B, et al. Hemoglobin concentration does not impact 3-month outcome following acute ischemic stroke. BMC Neurol 2018;18:78. [Crossref] [PubMed]
- Bhatia RS, Garg RK, Gaur SP, et al. Predictive value of routine hematological and biochemical parameters on 30-day fatality in acute stroke. Neurol India 2004;52:220-3. [PubMed]
- Czlonkowska A, Ryglewicz D, Lechowicz W. Basic analytical parameters as the predictive factors for 30-day case fatality rate in stroke. Acta Neurol Scand 1997;95:121-4. [Crossref] [PubMed]
- Bots ML, Nikitin Y, Salonen JT, et al. Left ventricular hypertrophy and risk of fatal and non-fatal stroke. EUROSTROKE: a collaborative study among research centres in Europe. J Epidemiol Community Health 2002;56 Suppl 1:i8-13. [Crossref] [PubMed]
- Tardif JC. Insights into oxidative stress and atherosclerosis. Can J Cardiol 2000;16 Suppl D:2D-4D.
- Sakanaka M, Wen TC, Matsuda S, et al. In vivo evidence that erythropoietin protects neurons from ischemic damage. Proc Natl Acad Sci U S A 1998;95:4635-40. [Crossref] [PubMed]
- Astor BC, Muntner P, Levin A, et al. Association of kidney function with anemia: the Third National Health and Nutrition Examination Survey (1988-1994). Arch Intern Med 2002;162:1401-8. [Crossref] [PubMed]
- Zhang L, Wang F, Wang L, et al. Prevalence of chronic kidney disease in China: a cross-sectional survey. Lancet 2012;379:815-22. [Crossref] [PubMed]
- Tsai TH, Lu CH, Wallace CG, et al. Erythropoietin improves long-term neurological outcome in acute ischemic stroke patients: a randomized, prospective, placebo-controlled clinical trial. Crit Care 2015;19:49. [Crossref] [PubMed]
- Kang J, Park J, Lee JM, et al. The effects of erythropoiesis stimulating therapy for anemia in chronic heart failure: A meta-analysis of randomized clinical trials. Int J Cardiol 2016;218:12-22. [Crossref] [PubMed]


