
Effect of chromatic contrast on stereoacuity measurement with computer-aided three-dimensional technology
Introduction
Stereoacuity is an important index for evaluating binocular function during an ophthalmic examination. Various measurement tools are used to detect the stereopsis threshold in the clinic, including the Frisby (1-3), The Netherlands Optical Society (TNO) (4-6), Titmus (7-9), and other stereoacuity tests. None of these test methods involve color information. Although the test picture of TNO comprises red and green colors, the purpose is to divide the two eyes’ images with green and red glasses, rather than investigating the effect of colors.
Computer-aided, three-dimensional (3D) evaluation systems have become more practical and have been utilized in important studies (10-15). The customizability of the test system is one of its greatest advantages over traditional stereoacuity threshold measurements. Incorporating colorful elements into test targets could help the test appear more vivid and attractive. However, it is unknown whether different color variations affect the stereopsis test results. In this investigation, we aimed to address whether different chromatic pair stereo targets would affect the stereo test results.
Methods
Computer system
The 3D computer system and software were set up as in our previous study (15). The experiment used a 3D laptop (ASUS G750Y47JX) and liquid crystal shutter glasses [NVidia 3D Vision 2 Wireless Glasses Kit (Expressway Santa Clara, CA, USA)]. The laptop was warmed up for 1 hour prior to measurement, and a color calibration instrument (Spyder 5 EXPRESS, Datacolor, Lawrenceville, NJ, USA) was used to calibrate the laptop monitor. A screen luminance meter (SM208, M&A Instrument Inc. Shenzhen, China) was used to measure the brightness of the display.
Test targets
A program written in C# programming language was used to produce all test targets. A square symbol (□) with a stroke width of 9 pixels (px) and a total size of 45×45 px was designed as the test symbol. A test page contained three rows, and each row had seven symbols, 100 px apart from each other, both horizontally and vertically. The stereo target was one symbol in a row randomly chosen from the five middle symbols and would be rendered at a different depth. The crossed disparity was used in this test. The reference stereo targets were set at 3 px in the top line and 2 px in the bottom line; the measurement stereo target, with the disparity of 1 px, was located in the middle line (Figure 1). The contrast between the symbols and background was calculated using the Weber contrast formula: C= [Imax-Imin]/Imin. Three channels including red (R), green (G), and blue (B) are involved in constituting the chromatic system.
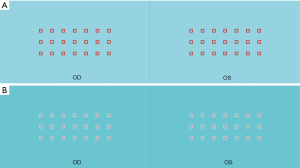
Maximum color contrast pages
We set up seven maximum color contrast pages (test pages 1 to 7), as shown in Figure 1. The specific parameters are shown in Table 1.
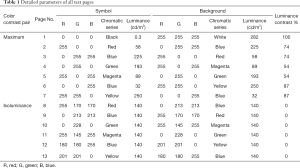
Full table
Isoluminant color contrast pages
We established six pages (test pages 8 to 13) for which the luminance equaled 140 cd/m2, approximately half of the maximum luminance of our system. Thus, the contrast was zero for each page (Figure 1, Table 1).
Test procedure
We used our variable distance test system to test stereoacuity. This method was detailed in our published manuscript (15). The subject sat at a relatively long distance from the screen—approximately 5 m—where the subject could not detect the stereo target on the middle line. The participant was then asked to slowly move toward the monitor until he or she could just point out the target symbol. At that point, the distance from the eyes to the display was measured and recorded with a laser rangefinder (GLM 30 Professional, BOSCH, Milton Keynes, UK). The subject was then asked to slowly move away from the monitor until he or she could no longer point out the target symbol, and the distance from the eyes to the display was measured. The stereoacuity was calculated using the formula incorporating distance and disparity: dγ=aΔd/d2 (16), where d was calculated as the mean distance between proximal and distal results.
Subjects
A total of 17 subjects (7 males and 10 females), aged 20 to 28 years, were recruited. None of the study participants had amblyopia, strabismus, anisometropia, or severe ametropia. The best corrected visual acuity was at least 0 logarithmic minimum angle of resolution (logMAR) for each eye. The color vision was normally tested with the pseudoisochromatic plate test (17). The stereoacuity was at least 32 second of arc (arcsec) measured by the Fly Stereo Acuity Test (Vision Assessment Corporation, Elk Grove Village, IL, USA).
All participants gave their informed written consent before taking part. The research protocol observed the tenets of the Declaration of Helsinki and was approved by the ethics committee of the Second Hospital of Jilin University (No. 2017-89).
Statistics
All data were processed using PASW Statistics 18 software (IBM SPSS Inc., Chicago, IL, USA). We first used the Shapiro-Wilk test to explore the distribution of data. We then chose parametric tests [one-way analysis of variance (ANOVA) test, paired-t-test] or non-parametric tests (Kruskal-Wallis test) to analyze the data based on the normality test result.
Results
Seventeen participants completed 13 experiments corresponding to 13 test pages, and the stereoacuity results are presented in Figure 2. The statistical analysis was conducted with distance rather than stereoacuity because distance data had a normal distribution, according to the Shapiro-Wilk test (P>0.05 in all 13 groups). One-way ANOVA was used to test the data. There was no significant difference found among the seven experiments of the maximum color contrast pairs (six test groups and one reference group, F=0.995, P=0.493). We also found no significant differences among the isoluminant color contrast pairs (six test groups, F=0.873, P=0.644).
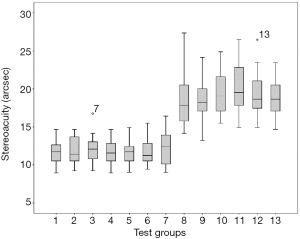
We used paired t-tests to compare data between the same hue series in maximum color contrast pairs vs. isoluminant color contrast pairs. We found significant differences in all six pairs. The detailed results were as follows: pair one, red symbol with light blue background, t=6.330, P<0.001; pair two, light blue symbol with red background, t=10.375, P<0.001; pair three, green symbol with magenta background, t=11.015, P<0.001; pair four, magenta symbol with green background, t=13.204, P<0.001; pair five, blue symbol with yellow background, t=11.252, P<0.001; pair six, yellow symbol with blue background, t=10.440, P<0.001.
Discussion
The relationship between chromatic information and stereopsis has been explored for several decades, and many controversial issues still puzzle researchers. The nerve system that processes chromatic components, luminance, and stereoacuity may involve the parvocellular and magnocellular pathways (18). The processing of stereoscopic depth information likely involves three stereopsis mechanisms: first- and second-order luminance stereopsis and first-order chromatic stereopsis (19,20).
While we did not elucidate the profound mechanism of chromatic or luminance contrast stereopsis, we solved a practical problem: Is it possible to incorporate colorful components into our 3D-computer assisted stereoacuity threshold test system? We evaluated the possibility of using chromatic test symbols and backgrounds to make the measurement system more vivid and interesting. Moreover, the colorful stereo test pattern we introduced into the system should not affect the results when compared with traditional stereo test symbols. The display mode of our 3D laptop was quite different from analogous tests to detect chromatic and luminance contrast and stereoacuity. The computer monitors used by researchers in the past were cathode ray tube (CRT) monitors, while today we use light-emitting diode (LED), monitors. Researchers have obtained satisfactory results using an autostereoscopic tablet to test stereopsis and establishing stimuli by importing color elements as random dots (21). However, the test design and the display screen in that system were far different from ours.
The chromatic system we used involved three channels: R, G, and B. Each channel had an integer value ranging from 0 to 255 inclusive and added together to represent one color. The display can, therefore, show 2553≈16.58 million distinct colors, from pure black (R =0, G =0, B =0) to pure white (R =255, G =255, B =255). R =255, G =0, and B =0 represent red, while R =0, G =255, and B =255 is the complementary color of red—i.e., light blue. The same scheme applies to other pairs of complementary colors, such as green (R =0, G =255, B =0) vs. magenta (R =255, G =0, B =255) and blue (R =0, G =0, B =255) vs. yellow (R =255, G =255, B =0). If the R channel stays at 255 while the other two channels increase at the same rate, then the red becomes lighter, and the color series remains in the red spectrum. If the G and B channels remain at 0 while the R channel decreases from 255, then the red becomes darker as the value decreases. We used this method to control the luminance of each color we tested in this experiment to gain the isoluminant test with the screen luminance meter.
In the maximum color contrast pairs experiment, none of the complementary color pairs showed a significant difference with the luminance Weber contrast ranging from 54% to 87%. We found no significant difference between the pairs in the isoluminant color contrast test. However, stereoacuity was significantly decreased in the isoluminant group when compared with the maximum color contrast group, which matched findings of prior studies (22-26). Some researchers believe that blue cones are absent in the fovea and are much less dense than red or green cones in the rest of the retina (27) and that this may affect the stereoacuity of isoluminant blue-on-yellow random-dot stereograms (28). However, this did not happen in our experiment.
Researchers have studied the effects of luminance contrast on stereopsis without color information using computer-aided stereoacuity tests. Although the details differ, the overall trend is consistent—that is, the threshold of stereopsis is quite stable at relatively high contrast conditions and changes dramatically at low contrast conditions. Legge et al. (29) found a correlation between disparity sensitivity and contrast sensitivity (r=0.84), and threshold disparity was inversely proportional to the square root of contrast. The researchers used vertical sine wave gratings to evaluate seven subjects’ stereopsis with different contrasts. The stereo targets were generated digitally with an LSI-11/23 computer and using a CRT monitor comprising four panels. Halpern et al. (30) investigated the relation between stereoacuity and contrast with a pair of matched CRT monitors (P31 phosphor) and a mirror stereoscope. The stimuli were the tenth derivatives of a Gaussian function. The investigators found that stereoacuity was independent of contrast at high-contrast conditions while the model varied in proportion to the square of the contrast at low-contrast conditions. Cormack et al. (31) explored the problem with a pair of matched touchscreen display monitors (P4 phosphor, 60 Hz noninterlaced) viewed through a mirror haploscope, and the stereo targets were dynamic random-element stereograms of 50% element density. Correlation thresholds were found to be independent of contrast at high contrasts, while correlation thresholds were inversely proportional to the square of contrast at low contrasts. In our experiment, the range of contrast in maximum color-contrast pairs (Weber contrast: 54–87%) was not low enough to affect the threshold of stereopsis. We did not consider other contrast conditions because the scope was overly complex for this study; moreover, that was not the emphasis of our study.
In practice, chromostereopsis (27,32,33), a type of color depth sense caused by color with a different wavelength, should not be neglected. If the red light is specifically focused on the retina from a certain distance, then blue light emitted from the same distance would focus in front of the retina plane, the shorter wavelength of the blue light may be expressed as a blur. This effect is known as longitudinal chromatic aberration (34). The light should be refracted by the refractive medium of the eye, but the refractive power changes with different wavelength. The angle alpha (angle between the optic axis and the visual axis through the fovea) aggravates the effect, especially in binocular viewing situations. This effect is known as transverse chromatic aberration (35). For example, if a small blue object and a small red object are placed vertically in the same frontal plane, crossed disparity for the red object against the blue object in the two eyes should be induced for a majority of subjects, causing the red object to appear nearer than the blue object. However, for subjects in whom the angle alpha has the opposite sign, there is an uncrossed disparity, and the blue object appears to be nearer than the red object (27). In our experiment, chromostereopsis existed for all symbols, so determining the outstanding symbol depended only on the disparities we set; results were not affected by chromostereopsis. If a stereopsis test design involves complex color matching, chromostereopsis should be considered.
One limitation of our research is that the participants were all young adults because of the complex stereopsis test procedure. Different populations, especially children, should be recruited for future tests using a simpler test protocol. The colors involved in the test were limited, and luminance contrast data was restricted. More color and luminance contrast should be considered in future studies. Another limitation is the lack of rigorous wavelength calibration, which reduces the potential for use in theoretical research into the effects of chromatic contrast on stereoacuity. However, the results of this study are acceptable for clinical applications, particularly for clinical screening.
Conclusions
Adding chromatic factors to a stereo test is practical when keeping the luminance contrast at a maximum level and maintaining the color contrast in a complementary manner. The exact threshold where luminance contrast begins to affect the results of stereo tests and the effects of color contrast require further study.
Acknowledgements
Funding: This study was supported by a grant from the Jilin Provincial Science & Technology Department, China (No. 20170519004JH, No. 20190303150SF).
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: All adult participants gave their informed written consent before taking part. For all underage participants, informed consent was obtained from the guardian before participation in the study. The research protocol observed the tenets of the Declaration of Helsinki and was approved by the ethics committee of The Second Hospital of Jilin University (No. 2017-89).
References
- Bohr I, Read JC. Stereoacuity with Frisby and revised FD2 stereo tests. PLoS One 2013;8:e82999. [Crossref] [PubMed]
- Costa MF, Moreira SM, Hamer RD, et al. Effects of age and optical blur on real depth stereoacuity. Ophthalmic Physiol Opt 2010;30:660-6. [Crossref] [PubMed]
- Anketell PM, Saunders KJ, Little JA. Stereoacuity norms for school-age children using the Frisby stereotest. J AAPOS 2013;17:582-7. [Crossref] [PubMed]
- Garnham L, Sloper JJ. Effect of age on adult stereoacuity as measured by different types of stereotest. Br J Ophthalmol 2006;90:91-5. [Crossref] [PubMed]
- Lee JY, Seo JY, Baek SU. The effects of glasses for anisometropia on stereopsis. Am J Ophthalmol 2013;156:1261-6.e1. [Crossref] [PubMed]
- van Doorn LL, Evans BJ, Edgar DF, et al. Manufacturer changes lead to clinically important differences between two editions of the TNO stereotest. Ophthalmic Physiol Opt 2014;34:243-9. [Crossref] [PubMed]
- Tejedor J, Ogallar C. Comparative efficacy of penalization methods in moderate to mild amblyopia. Am J Ophthalmol 2008;145:562-9. [Crossref] [PubMed]
- Yang JW, Son MH, Yun IH. A study on the clinical usefulness of digitalized random-dot stereoacuity test. Korean J Ophthalmol 2004;18:154-60. [Crossref] [PubMed]
- Arnoldi K, Frenkel A. Modification of the titmus fly test to improve accuracy. Am Orthopt J 2014;64:64-70. [Crossref] [PubMed]
- Han SB, Yang HK, Kim J, et al. New stereoacuity test using a 3-dimensional display system in children. PLoS One 2015;10:e0116626. [Crossref] [PubMed]
- Jung JH, Yeom J, Hong J, et al. Effect of fundamental depth resolution and cardboard effect to perceived depth resolution on multi-view display. Opt Express 2011;19:20468-82. [Crossref] [PubMed]
- Han J, Han SY, Lee SK, et al. Real stereopsis test using a three-dimensional display with Tridef software. Yonsei Med J 2014;55:1672-7. [Crossref] [PubMed]
- Ma DJ, Yang HK, Hwang JM. Reliability and validity of an automated computerized visual acuity and stereoacuity test in children using an interactive video game. Am J Ophthalmol 2013;156:195-201.e1. [Crossref] [PubMed]
- Kim J, Yang HK, Kim Y, et al. Distance stereotest using a 3-dimensional monitor for adult subjects. Am J Ophthalmol 2011;151:1081-6.e1. [Crossref] [PubMed]
- Wu H, Jin H, Sun Y, et al. Evaluating stereoacuity with 3D shutter glasses technology. BMC Ophthalmol 2016;16:45. [Crossref] [PubMed]
- Saladin JJ. Phorometry and Stereopsis. In: Benjamin WJ. editor. Borish's Clinical Refraction. 2nd Edition. St. Louis: Butterworth–Heinemann Elsevier, 2006:899-960.
- Yu Ziping, Cao Yu, Cao Kai, editors. Color vision examination plates. 6th Edition. Beijing: People’s Medical Publishing House, 2017.
- Howard IP, Rogers BJ. Binocular vision and stereopsis. New York: Oxford University Press, 1995.
- Simmons DR, Kingdom FA. Interactions between chromatic- and luminance-contrast-sensitive stereopsis mechanisms. Vision Res 2002;42:1535-45. [Crossref] [PubMed]
- Simmons DR, Kingdom FA. On the binocular summation of chromatic contrast. Vision Res 1998;38:1063-71. [Crossref] [PubMed]
- Serrano-Pedraza I, Vancleef K, Read JC. Avoiding monocular artifacts in clinical stereotests presented on column-interleaved digital stereoscopic displays. J Vis 2016;16:13. [Crossref] [PubMed]
- Simmons DR, Kingdom FA. Differences between stereopsis with isoluminant and isochromatic stimuli. J Opt Soc Am A Opt Image Sci Vis 1995;12:2094-104. [Crossref] [PubMed]
- Simmons DR, Kingdom FA. Contrast thresholds for stereoscopic depth identification with isoluminant and isochromatic stimuli. Vision Res 1994;34:2971-82. [Crossref] [PubMed]
- Kingdom FA, Simmons DR. Stereoacuity and colour contrast. Vision Res 1996;36:1311-9. [Crossref] [PubMed]
- Troscianko T, Montagnon R, Le Clerc J, et al. The role of colour as a monocular depth cue. Vision Res 1991;31:1923-9. [Crossref] [PubMed]
- Osuobeni EP, O'Leary DJ. Chromatic and luminance difference contribution to stereopsis. Am J Optom Physiol Opt 1986;63:970-7. [Crossref] [PubMed]
- Howard IP. Perceiving in Depth. New York: Oxford University Press, 2012.
- Grinberg DL, Williams DR. Stereopsis with chromatic signals from the blue-sensitive mechanism. Vision Res 1985;25:531-7. [Crossref] [PubMed]
- Legge GE, Gu YC. Stereopsis and contrast. Vision Res 1989;29:989-1004. [Crossref] [PubMed]
- Halpern DL, Blake RR. How contrast affects stereoacuity. Perception 1988;17:483-95. [Crossref] [PubMed]
- Cormack LK, Stevenson SB, Schor CM. Interocular correlation, luminance contrast and cyclopean processing. Vision Res 1991;31:2195-207. [Crossref] [PubMed]
- Vos JJ. Depth in colour, a history of a chapter in physiologie optique amusante. Clin Exp Optom 2008;91:139-47. [Crossref] [PubMed]
- Ozolinsh M, Muizniece K. Color difference threshold of chromostereopsis induced by flat display emission. Front Psychol 2015;6:337. [Crossref] [PubMed]
- Wang J, Candy TR, Teel DF, et al. Longitudinal chromatic aberration of the human infant eye. J Opt Soc Am A Opt Image Sci Vis 2008;25:2263-70. [Crossref] [PubMed]
- Faubert J. Colour induced stereopsis in images with achromatic information and only one other colour. Vision Res 1995;35:3161-7. [Crossref] [PubMed]


