The usefulness of transient elastography in patients with autoimmune hepatitis—when can we use it?
Recently Hartl et al. (1,2) investigated the diagnostic performance of transient elastography (TE) in patients with autoimmune hepatitis (AIH). They found that TE has an excellent diagnostic accuracy for separating severe from non-severe fibrosis in AIH. These findings are very interesting but should be interpreted with cautions as mentioned by the authors. Unfortunately, liver biopsy is still the gold standard to diagnose liver fibrosis (3). However, it has many drawbacks, such as sampling error, risk of complications, etc. Therefore, there is great interest in determining non-invasive markers for the diagnosis of liver fibrosis.
As we know, TE has demonstrated good accuracy to diagnose advanced fibrosis and cirrhosis (F3–F4), independent of the underlying etiology (4), showing an average of sensitivity, specificity and area under the ROC curve about 0.9, 0.8 and 0.92, respectively (5-7). Moreover, the sampling volume of TE is bigger than that of liver biopsy and it has been suggested, consequently, that the results of TE are more representative of the hepatic parenchyma (8). However, TE has important limitations such as its reproducibility is significantly reduced in patients with steatosis, ascites or increased body mass index (9). Furthermore, the diagnostic performance of TE is particularly affected in patients with high serum levels of ALT such as patients with acute hepatitis or those with AIH (10).
On the other hand, Hartl et al. reported that TE shows high accuracy for diagnosing cirrhosis and severe fibrosis only after 6 months of immunosuppressive treatment (1,2). This result shows the impact of hepatic inflammation on liver stiffness and demonstrates that TE is not useful in the acute setting. Consequently, the diagnostic performance of TE to assess lower degrees of liver fibrosis (F0–F2) is very poor.
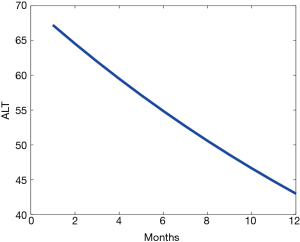
This leads us to ask the question, why the TE is inaccurate before this period of time? To answer the question, we have analyzed the results of the Hartl et al. (1,2) study, focusing on the reduction of ALT levels. It is possible to take characteristics of patients with different time intervals between start of treatment and TE measurement. The initial value of ALT was 70 U/L. After 6–18 months the ALT value decreased to 43 U/L. If the mean value of the Group 2 interval is taken, then it is reasonable to take 12 as the final time. If the premise of constant rate decrease of ALT is assumed, then the yearly rate is:
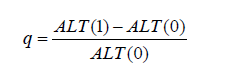
The monthly rate is easily derived and it comes out to be:
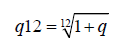
The constant-rate predictive expression for monthly units of time is:
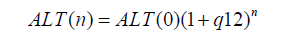
Figure 1 shows the ALT decay curve. According to the reduction of ALT levels, we suggest that the TE probably is useful in patients with AIH when the levels of ALT decreased with an intensive immunosuppressive treatment by 15%. In addition, the intensive treatment probably normalizes the ALT levels before 6 months, making earlier the diagnosis of the liver fibrosis in patients with AIH. However, more clinical studies are necessary to confirm our observations.
Acknowledgements
This study was supported in part by a grant from Medica Sur Clinic & Foundation.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Hartl J, Ehlken H, Sebode M, et al. Usefulness of biochemical remission and transient elastography in monitoring disease course in autoimmune hepatitis. J Hepatol 2017. [Epub ahead of print]. [PubMed]
- Hartl J, Denzer U, Ehlken H, et al. Transient elastography in autoimmune hepatitis: Timing determines the impact of inflammation and fibrosis. J Hepatol 2016;65:769-75. [Crossref] [PubMed]
- Castera L, Pinzani M. Biopsy and non-invasive methods for the diagnosis of liver fibrosis: does it take two to tango? Gut 2010;59:861-6. [Crossref] [PubMed]
- Lucero C, Brown RS Jr. Noninvasive Measures of Liver Fibrosis and Severity of Liver Disease. Gastroenterol Hepatol (N Y) 2016;12:33-40. [PubMed]
- Huang R, Jiang N, Yang R, et al. Fibroscan improves the diagnosis sensitivity of liver fibrosis in patients with chronic hepatitis B. Exp Ther Med 2016;11:1673-7. [Crossref] [PubMed]
- Pathik P, Ravindra S, Ajay C, et al. Fibroscan versus simple noninvasive screening tools in predicting fibrosis in high-risk nonalcoholic fatty liver disease patients from Western India. Ann Gastroenterol 2015;28:281-6. [PubMed]
- El-Hariri MA, Abd El Megid AG, Taha Ali TF, et al. Diagnostic value of Transient Elastography (Fibroscan) in the evaluation of liver fibrosis in chronic viral hepatitis C: Comparison to liver biopsy. Egyptian Journal of Radiology and Nuclear Medicine 2017;48:329-37. [Crossref]
- Uribe-Ramos M, Méndez-Sánchez N. Why is transient elastography essential in hepatology? Ann Hepatol 2009;8:148-9. [PubMed]
- Juárez-Hernández E, Uribe-Ramos MH, Ramos-Ostos MH, et al. Factors Associated with the Quality of Transient Elastography. Dig Dis Sci 2015;60:2177-82. [Crossref] [PubMed]
- Chang PE, Goh GB, Ngu JH, et al. Clinical applications, limitations and future role of transient elastography in the management of liver disease. World J Gastrointest Pharmacol Ther 2016;7:91-106. [Crossref] [PubMed]