Blood pressure targets for hypertension in patients with type 2 diabetes
Introduction
One of the main goals in managing type 2 diabetes in adults is prevention of cardiovascular morbidity or mortality by controlling blood glucose, normalizing blood pressure, and reducing other cardiovascular risk factors (1,2). Despite extensive review of literature, clinical practice guidelines vary in determining the optimal blood pressure targets in patients with diabetes (3-5).
To support clinical decisions at point of care with all available evidence, we conducted a rapid review of the published and unpublished data from recently completed randomized controlled trials (RCT), meta-analyses of RCTs, and primary observational studies that compared different blood pressure targets in adults with diabetes.
Methods
We used a standard recommended methodology in conducting systematic literature reviews and meta-analyses from the Cochrane Collaboration and the Agency for Healthcare Research and Quality (6,7). We developed a priori protocol (available by request) for a systematic literature review to answer the clinical question about the comparative effectiveness of blood pressure targets on mortality and cardiovascular morbidity in adults with diabetes mellitus.
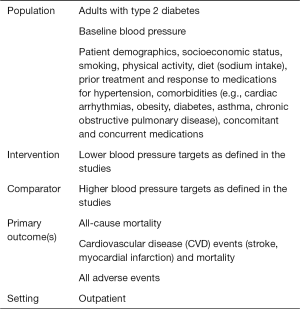
Eligible studies directly compared lower versus higher blood pressure targets or examined the association between baseline or attained blood pressure with patient outcomes in people treated with hypotensive medications. Eligible outcomes included all-cause and underlying cause-specific mortality, cardiovascular morbidity, stroke, heart failure, renal failure, and all drug harms.
We conducted a comprehensive search in PubMed, EMBASE, the Cochrane Library, and www.clinicaltrials.gov up to March 2018 to find systematic reviews, published and unpublished RCTs, and nationally represented controlled observational studies that reported adjusted effect estimates (6,7). The data were extracted from the Clinical Trials Transformation Initiative (https://www.ctti-clinicaltrials.org/aact-database), checked for quality, and stored in the High-Performance Computing Cluster platform (https://hpccsystems.com).
We tested the null hypotheses of no differences in patient outcomes after more versus less extensive blood pressure lowering (6). We abstracted the information about study population, interventions, comparators, and outcomes (6). We abstracted minimum datasets (e.g., number of the subjects in treatment groups and events) to estimate absolute risk difference, relative risk, and number needed to treat for categorical variables (6). Statistical significance was evaluated at a 95% confidence level (including the use of P values).
We conducted a rapid review following the framework of the AHRQ (8). We used the AHRQ recommended methodological approach in the integration of existing systematic reviews into our comprehensive synthesis of evidence (9). Our goal was the integration of previously published high quality reviews and consistent ranking of the quality of evidence using GRADE methodology.
We performed meta-analyses when definitions of active and control interventions and patient outcomes deem similar (10). We examined consistency in results across studies with chi-square tests and I2 statistics and concluded statistically significant heterogeneity if I2 was >50% (6). Statistically significant heterogeneity did not preclude statistical pooling (10). However, we planned exploring heterogeneity with a priori defined patient baseline hypertensive status (10).
We defined harms as the totality of all possible adverse consequences of an intervention.
We calculated absolute risk difference, number needed to treat, and the number of attributable events based on data from the published randomized trials, using STATA software (StataCorp LP, College Station, TX, USA) (11). Correction coefficients for zero events were used as a default option, and intention to treat was used for evidence synthesis (10). Superiority of interventions under comparison was hypothesized (12). We used consensus method guidelines for systematic review and meta-analyses that do not recommend conducting post hoc analyses of statistical power (13-15). Instead, we downgraded our confidence in true treatment effects based on calculated optimal information size as the number of patients required for an adequately powered individual trial (16). Since power is more closely related to number of events than to sample size, we concluded imprecision in treatment effects if less than 250 patients experienced the event (16).
We assessed reporting bias as a proportion of published among all registered studies, unreported outcomes compared with published protocols, or unreported minimum data sets for reproducibility of the results (17). We did not conduct formal statistical tests for publication bias due to the questionable validity of such tests (18).
We evaluated the quality of the primary studies using the Cochrane risk of bias tool on a 3-point scale: high bias, low bias, and unclear (6). We upgraded the risk of bias in the body of evidence from low to high if at least 1 RCT had high risk of bias (19,20). We defined indirectness in outcomes from intermediate outcomes (21).
Treatment effect estimates were defined as precise when pooled estimates had reasonably narrow 95% confidence intervals (CI) and the number of events were greater than 250 (22). Justification of the sample size was not included in grading of the evidence.
In assessing the quality of evidence in all studies, the authors looked for the strength of association and evidence of any reporting bias (23). The strength of the association was evaluated, defining a priori a large effect when the relative risk was greater than 2 and a very large effect when the relative risk was greater than 5 (23). A small treatment effect was construed when the relative risk was significant but less than 2 (23).
The authors assigned the quality of evidence ratings as high, moderate, low, or very low, according to risk of bias in the body of evidence, directness of comparisons, precision and consistency in treatment effects, and the evidence of reporting bias, using Grading of Recommendations Assessment, Development and Evaluation (GRADE) methodology (Supplementary file) (23). A high quality of evidence was assigned to well-designed RCTs with consistent findings. The quality of evidence was downgraded to moderate if at least 1 of 4 quality of evidence criteria were not met; for example, moderate quality of evidence was assigned if there was a high risk of bias in the body of evidence or if the results were not consistent or precise (23). The quality of evidence was downgraded to low if 2 or more criteria were not met.
A low quality of evidence was assigned to nonrandomized studies and upgraded for the rating if there was a strong association. Evidence was defined as insufficient when no studies provided valid information about treatment effects. This approach was applied regardless of whether the results were statistically significant.
Results
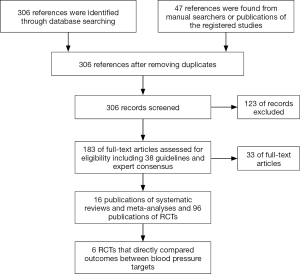
Our comprehensive search in PubMed, EMBASE, the Cochrane Library, and clinicaltrials.gov up to March 2018 retrieved 306 references and identified 16 publications of systematic reviews and meta-analyses and 96 publications of RCTs that enrolled adults primarily with type 2 diabetes mellitus (Supplementary file, Figure S1). We excluded 124 irrelevant references at the screening of the titles and abstracts and 33 publications after full text review because the studies did not address blood pressure targets in patients with diabetes (Supplementary file, Figure S1). Only 6 primary RCTs randomly assigned patients to lower versus higher blood pressure targets and compared systolic blood pressure (SBP) targets in patients with baseline arterial hypertension (24,25) or diastolic blood pressure (DBP) targets in patients with normal (26,27) or elevated baseline blood pressure (28,29). All other RCTs compared hypotensive drugs with placebo or with each other. Meta-analyses of such trials explored the association of baseline or achieved blood pressure with patient outcomes (3-5,30-39).
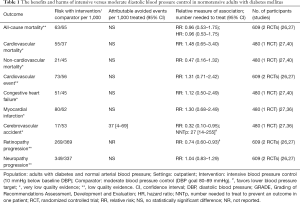
In adults with diabetes and normal arterial blood pressure, low-quality evidence suggests that there are no differences in all-cause and cardiovascular mortality or morbidity between intensive (10 mmHg below baseline DBP) and moderate blood pressure control (DBP goal 80–89 mmHg; Table 1). Intensive blood pressure control prevents cerebrovascular events and progression or retinopathy in some patients (Table 1).
Full table
In adults with diabetes and elevated arterial blood pressure (DBP ≥90 mmHg), low-quality evidence suggests that there are no differences in all-cause and cardiovascular mortality or stroke between intensive (DBP ≤85–75 mmHg) and moderate blood pressure control (DBP goal 80–90 mmHg; Table 2). A single RCT suggests that a reduction of DBP ≤80 mmHg results in a lower risk of major cardiovascular events but higher risk of progressing neuropathy (Table 2).
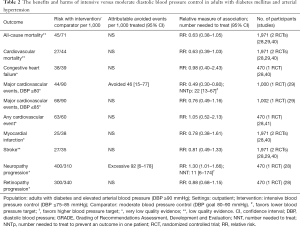
Full table
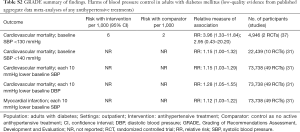
In adults with diabetes and elevated arterial blood pressure (SBP 130–190 mmHg), moderate-quality evidence suggests that there are no differences in all-cause or cardiovascular mortality between intensive and standard blood pressure control (Table 3 and Figure 1). However, intensive blood pressure control decreases the risk of diabetes-related mortality, fatal or nonfatal stroke (Figure 2), prevalence of left ventricular hypertrophy and electrocardiogram (ECG) abnormalities, macroalbuminuria, and non-spine bone fractures (Table 3). A single RCT (ACCORD) suggests that the risk of a composite outcome (nonfatal myocardial infarction, nonfatal stroke, and death from cardiovascular causes) is lower after intensive blood pressure and good glycemic control but higher in adults with poorly controlled diabetes (hemoglobin A1c >8.0; Table 3). The benefits from intensive blood pressure control sustain at 9 years of follow-up in older adults with 15% or greater 10-year coronary heart risk in the standard glucose control arm of ACCORD trial (Table 3). The same study reported an increased risk of adverse effects from hypotensive medications, including hypotension or hyperkalemia, after intensive blood pressure control (Table 3).
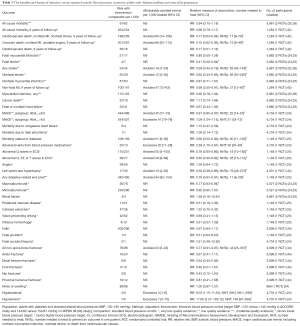
Full table
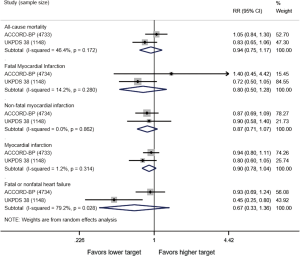
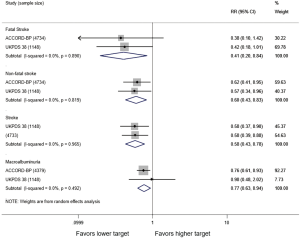
Primary studies did not address circadian fluctuations in blood pressure or the risk of orthostatic hypotension after intensive versus standard blood pressure targets. For the record, the ACCORD study found no differences in patient falls or trauma after intensive blood pressure control (Table 3).
Published meta-analyses of aggregate data from RCTs aimed at efficacy or comparative effectiveness of specific hypotensive drugs in adults with comorbid diabetes and arterial hypertension (baseline blood pressure >150 mmHg) agree that attained SBP 130–139 and DBP 75–80 mmHg is associated with lower risk of all-cause mortality and stroke (Table S1) (5,31,33,37). Systematic reviews and guidelines vary in determining the balance between benefits and harms of lowering blood pressure below 130 mmHg (4,5). We looked at the pooled relative risk of all reported outcomes in published reviews (Table S1) and found 2 meta-analyses that reported a statistically significant increase in the risk of cardiovascular mortality in association with lower baseline blood pressure (Table S2) (31,37). Anti-hypertensive treatments are associated with a higher risk of cardiovascular mortality per each 10 mmHg lower baseline SBP and DBP (Table S2).

Full table
Full table
Discussion
Our review found moderate direct quality evidence that in adults with diabetes and elevated SBP, intensive blood pressure control (target SBP <120–140 mmHg) decreases the risk of diabetes-related mortality, fatal or nonfatal stroke, prevalence of left ventricular hypertrophy and ECG abnormalities, macroalbuminuria, and non-spine bone fractures, with no differences in all-cause or cardiovascular mortality or falls.
We downgraded the quality of evidence due to risk of bias, small number of events in the studies, and heterogeneity in treatment effects across the studies. We did not conduct meta-regression of few RCTs that directly compared patient outcomes after intensive versus standard blood pressure targets (6). Instead, we reviewed published meta-analyses of RCTs aimed at comparative effectiveness of blood lowering drugs in adults with diabetes that concluded no benefits from lowering blood pressure below 130 mmHg. Such publications employed meta-regression of aggregated data that can generate hypotheses of potential harms from extensive lowering blood pressure control specifically in adults with normal baseline blood pressure (45). Published meta-analyses that included the data from the Systolic Blood Pressure Intervention Trial (SPRINT) suggest similar reduction in the risk of major cardiovascular events from intensive blood pressure lowering in adults with and without diabetes (20,38,46). Individual rather than aggregate patient data meta-analyses would provide better evidence about the association between specific drugs, baseline and achieved blood pressure, and patient outcomes independent of drug effects (47).
We found no studies that addressed the risk of hospitalization or long-term quality of life in relation to blood pressure targets in adults with diabetes. Although orthostatic hypotension is associated with poor patient outcomes, the evidence regarding the risk of this complication after intensive or standard blood pressure control is insufficient (3,48,49). Primary studies and meta-analyses did not discuss the importance of pulse pressure in reducing morbidity and mortality in adults with diabetes (50).
Guidelines recommend healthy diet, weight normalization, and physical activity for all adults with diabetes (Table S3) (51-58).

Full table
In addition to healthy lifestyle, recent guidelines recommend antihypertensive drug treatments in patients with diabetes and baseline blood pressure ≥130/80 mmHg (54-56,59-61). The American Heart Association recommends a treatment goal of <130/80 mmHg, while the American Diabetes Association recommends a treatment goal of <140/90 mmHg with lower targets in individuals at high risk of cardiovascular disease (54-56,59-61). Other guidelines also recommend baseline cardiovascular disease risk assessment and evaluation of kidney, eye, or cerebrovascular damage in determining individual treatment goals (51,62-65). The American Geriatrics Society guidelines acknowledge the potential harm from arterial hypotension in older adults with diabetes mellitus (66). Guidelines generally agree that high quality care for patients with diabetes include normalization of HbA1C without hypoglycemia (67-69). This definition of high quality care for patients with diabetes should include normalization of blood pressure including pulse pressure without hypotension.
Regardless of the intended blood pressure goal, the ability to maintain a lower blood pressure threshold in a real-world setting outside of a controlled trial is an important disease management consideration (70). It is often reported that more than half of treated patients are not able to maintain blood pressure control, even at a threshold of <140/85 mmHg (71). Findings from the DIALOGUE study (72), a multicenter prospective registry among patients with hypertension and type II diabetes, demonstrated that patients with a “strict” SBP target (≤130 mmHg) had more contacts with general practitioners than any other patient group. In addition, among patients with a lower blood pressure target, only half actually maintained this threshold over 6 months. More specifically, 53% of patients in the “strict” target group (≤130 mmHg) were able to maintain this blood pressure goal over time, and 55% of patients in the “medium” target group (130 to ≤135 mmHg) were able to do so.
The majority of the studies relied on office measurements of blood pressure rather than ambulatory blood pressure monitoring. However, ambulatory blood pressure monitoring improves baseline and post-treatment risk assessment (73-86). Evidence-based guidelines recommend ambulatory blood pressure monitoring for diagnosis and individualization of treatment goals in adults with arterial hypertension (51,56,87-89).
Our review has several limitations. We analyzed direct evidence from RCTs that randomly assigned patients to more versus less intensive blood pressure goals and did not abstract the data from RCTs aimed at efficacy or comparative effectiveness of hypotensive drugs. We could not reproduce the results from meta-regression, because the authors did not provide sufficient data (30,33,35-38,90). We did not contact authors of meta-analyses requesting reproducible data. We do not know how many unregistered and unpublished studies analyzed the association between baseline and attained blood pressure and patient outcomes.
Despite this limitation, we present conflicting evidence from all published and unpublished studies appraised with consistent GRADE methodology. In contrast with previous meta-analyses of direct evidence, we grouped studies by baseline hypertension status and by targeted diastolic and SBP targets (38,40).
Our review has implications for clinical practice. Clinicians should assess baseline cardiovascular risk, recommend behavioural and pharmacological treatments aiming at blood pressure normalization without hypotension or orthostatic hypotension (91). They should engage patients in life style optimization, blood pressure self-monitoring, and monitoring of drug adverse effects (92).
Our review has policy implications. High quality care in patients with diabetes and arterial hypertension should be defined as achievement of normal blood pressure without episodes of hypotension and with minimal risk of orthostatic hypotension or other serious harms from recommended drugs.
Our review has research implications. Future research should determine the optimal blood pressure targets in subpopulations with diabetes and various demographic, socioeconomic, and behavioral factors, as well as comorbidities. Composite outcomes should be avoided. Trials should use blood pressure monitoring and examine pulse pressure, the risk of orthostatic hypotension and other drug-related harms in determining optimal choice of drugs and blood pressure targets in individual patients.
Conclusions
Based on our review, we conclude that in adults with diabetes and arterial hypertension, in order to reduce the risk of stroke, left ventricular hypertrophy and ECG abnormalities, macroalbuminuria, and non-spine bone fractures, clinicians should encourage healthy lifestyle choices and antihypertensive medications targeting blood pressure of 120–130/80 mmHg, with close monitoring of daily blood pressure fluctuations, episodes of orthostatic hypotension, and other drug-related harms.
PICO question this report is addressing:
What are the benefits and harms of “lower” blood pressure targets compared to “standard” blood pressure targets in high-risk diabetic patients?
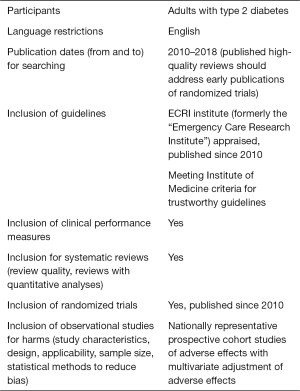
Study eligibility
Inclusion criteria
Exclusion criteria
Search strategy
The medical librarian develops specific search strategies based on the PICOs formulated by our clinical and epidemiology staff. We search for all relevant articles published in English from 2010 up to March 2018 in PubMed, EMBASE, and the Cochrane Library. To identify grey unpublished data, we conduct a search of the trial registry clinicaltrials.gov.
We conduct the following searches:
PubMed searches for:
- RCTs;
- Observational studies of harms (multivariate adjusted estimates from nationally representative cohorts or administrative databases) (6,7);
- Clinical practice guidelines.
EMBASE searches for full publications of:
- RCTs;
- Observational studies (multivariate adjusted estimates from nationally representative cohorts or administrative databases).
The bibliographies of identified articles are scanned, and study investigators are contacted for additional publications.
Study selection
The study epidemiologist and an author-subject matter expert contribute equally to resolving differences and decide the determination of eligibility collaboratively.
The study epidemiologist and an author-subject matter expert determine eligibility for full text review, first screen title, and abstracts. All citations found during the searches are stored in a reference database.
Data extraction and strategy for data synthesis
Data extraction
The data was extracted from the Clinical Trials Transformation Initiative (https://www.ctti-clinicaltrials.org/aact-database), checked for quality, and stored in the HPCC platform (High-Performance Computing Cluster, https://hpccsystems.com/).
We manually abstracted the data from published articles into the abstraction form. We checked the data for ambiguity (i.e., data reported in percentiles conflicting with unit data and vice versa; values outside a normal range) and mismatch with the published data. Identified errors have been discussed and corrected.
We abstract the information about study population, interventions, comparators, and outcomes. We abstract minimum datasets (e.g., number of the subjects in treatment groups and events) to estimate absolute risk difference, relative risk, and number needed to treat for categorical variables.
Means and standard deviations of continuous variables, e.g., total scores from the quality of life scales are abstracted. Statistical significance is evaluated at a 95% confidence level (including the use of P values). All authors have access to the data.
We conduct an overview of the reviews following the framework of the Cochrane Collaboration. We perform meta-analyses or update published meta-analyses. Pooling criteria include the exact same definitions of the active and control intervention, patient outcomes, and similar follow-up time (10).
We define harms as the totality of all possible adverse consequences of an intervention. Investigators sometimes defined harmful effects as unrelated to examined treatments. Harms are analyzed regardless of how investigators related them to treatments.
We calculate absolute risk difference, number needed to treat, and the number of attributable events based on data from the published randomized trials, using STATA software. Correction coefficients for zero events are used as a default option in both software programs, and intention to treat is used for evidence synthesis. Superiority of interventions under comparison is hypothesized.
We assess reporting bias as a proportion of published among all registered studies, unreported outcomes compared with published protocols, or unreported minimum data sets for reproducibility of the results. We did not conduct formal statistical tests for publication bias due to the questionable validity of such tests (18).
To examine the role of patient characteristics, a search is undertaken for subgroup analyses by patient demographics, baseline and achieved blood pressure, prior treatment response, and comorbidities in systematic reviews and randomized trials, including significant interaction effects.
Methodological assessment of the included studies
For systematic reviews (QIRs), we use the Assessment of Multiple Systematic Reviews (AMSTAR) scale to determine the methodological strength of the systematic reviews (99).
For randomized studies, we apply the Cochrane risk of bias tool. Risk of bias is assessed on a 3-point scale: high bias, low bias, and unclear (100,101). A low risk of bias is assumed when RCTs met all the risk-of-bias criteria, a medium risk of bias if at least 1 of the risk-of-bias criteria is not met, and a high risk of bias if 2 or more risk-of-bias criteria are not met. An unknown risk of bias is assigned for the studies with poorly reported risk-of-bias criteria. We assign high risk of bias to all observational studies.
For clinical practice guidelines, we use the Appraisal of Guidelines for Research and Evaluation (AGREE) II (2009) tool, which covers 23 items in 6 domains and 2 overall global ratings (102,103).
Quality assessment of the included studies and the body of evidence by outcome according to the GRADE framework
The authors assign the quality of evidence ratings as high, moderate, low, or very low, according to risk of bias in the body of evidence, directness of comparisons, precision and consistency in treatment effects, and the evidence of reporting bias, using Grading of Recommendations Assessment, Development and Evaluation (GRADE) methodology (23). We upgrade the risk of bias from low to high if at least 1 RCT had high risk of bias. We define indirectness in outcomes from intermediate outcomes. We review published network meta-analyses but do not conduct indirect comparisons.
Treatment effect estimates are defined as precise when pooled estimates have reasonably narrow 95% confidence intervals and the number of events are greater than 250. Justification of the sample size is not included in grading of the evidence. We do not conduct post hoc statistical power analyses.
In assessing the quality of evidence in all studies, the authors look for a dose response association, the strength of association, and evidence of any reporting bias. The strength of the association is evaluated, defining a priori a large effect when the relative risk is greater than 2 and a very large effect when the relative risk is greater than 5. A small treatment effect is construed when the relative risk was significant but less than 2. For standardized continuous measures of secondary and intermediate outcomes, the magnitude of the effect is defined according to Cohen et al. as small, moderate, and large, corresponding to mean differences in standard deviation units of 0 to 0.5, 0.5 to 0.8, and greater than 0.8, respectively.
A high quality of evidence is assigned to well-designed RCTs with consistent findings. The quality of evidence is downgraded to moderate if at least 1 of 4 quality of evidence criteria is not met; for example, moderate quality of evidence is assigned if there was a high risk of bias in the body of evidence or if the results are not consistent or precise. The quality of evidence is downgraded to low if 2 or more criteria are not met.
A low quality of evidence is assigned to nonrandomized studies and upgraded for the rating if there was a strong or dose-response association. Evidence is defined as insufficient when no studies provided valid information about treatment effects. This approach is applied regardless of whether the results were statistically significant.
The authors assign strength of the recommendations based on overall quality of evidence, balances between benefits and harms, healthcare consumers’ and clinicians’ values and preferences, and cost-effectiveness studies using the GRADE methodology.
PubMed search of record:
(((((((((((((((diabet*[Title/Abstract]) OR "Diabetes Mellitus/drug therapy"[Majr])) AND (((((hypertensi*[Title/Abstract]) OR (("Blood Pressure/drug effects"[Mesh] OR "Blood Pressure/pharmacology"[Mesh] OR "Blood Pressure/therapy"[Mesh]))) OR (((("systolic blood pressure"[Title/Abstract]) OR "systolic pressure"[Title/Abstract]) OR "diastolic blood pressure"[Title/Abstract]) OR "diastolic pressure"[Title/Abstract])) OR normotensive[Title/Abstract]) OR "Hypertension/drug therapy"[Majr]))) AND ((((((strict*[Title/Abstract]) OR target*[Title/Abstract]) OR tight*[Title/Abstract]) OR intens*[Title/Abstract]) OR below[Title/Abstract]) OR moderat*[Title/Abstract])))) AND (((((("Antihypertensive Agents"[Mesh]) OR "Antihypertensive Agents" [Pharmacological Action]) OR "Angiotensin II Type 1 Receptor Blockers"[Mesh])) OR antihypertensive[Title/Abstract]) OR "angiotensin II"[Title/Abstract]))) NOT ((((((("Letter"[Publication Type]) OR "News"[Publication Type]) OR "Patient Education Handout"[Publication Type]) OR "Comment"[Publication Type]) OR "Editorial"[Publication Type])) OR "Newspaper Article"[Publication Type]))) NOT (("Animals"[Mesh]) NOT (("Animals"[Mesh]) AND "Humans"[Mesh])))) AND (((((((((random*[Title/Abstract]) OR placebo*[Title/Abstract]) OR "double blind"[Title/Abstract]) OR "triple blind"[Title/Abstract]) OR prospective[Title/Abstract]) OR multicenter[Title/Abstract])) OR "Multicenter Study" [Publication Type]) OR "Randomized Controlled Trial" [Publication Type:NoExp])))
PubMed search of record for CPGs:
(((((((((((diabet*[Title/Abstract]) OR "Diabetes Mellitus/drug therapy"[Majr])) AND (((((hypertensi*[Title/Abstract]) OR (("Blood Pressure/drug effects"[Mesh] OR "Blood Pressure/pharmacology"[Mesh] OR "Blood Pressure/therapy"[Mesh]))) OR (((("systolic blood pressure"[Title/Abstract]) OR "systolic pressure"[Title/Abstract]) OR "diastolic blood pressure"[Title/Abstract]) OR "diastolic pressure"[Title/Abstract])) OR normotensive[Title/Abstract]) OR "Hypertension/drug therapy"[Majr]))) AND ((((((strict*[Title/Abstract]) OR target*[Title/Abstract]) OR tight*[Title/Abstract]) OR intens*[Title/Abstract]) OR moderat*[Title/Abstract])))) AND (((((("Antihypertensive Agents"[Mesh]) OR "Antihypertensive Agents" [Pharmacological Action]) OR "Angiotensin II Type 1 Receptor Blockers"[Mesh])) OR antihypertensive[Title/Abstract]) OR "angiotensin II"[Title/Abstract]))) AND (((((((((((((((((((((((("Guideline"[Publication Type]) OR "Practice Guideline"[Publication Type])) OR "Consensus Development Conference"[Publication Type]) OR "Consensus Development Conference, NIH"[Publication Type]) OR "Practice Guideline"[Publication Type]) OR "Guideline"[Publication Type]) OR ((clinical[Title]) AND guideline*[Title])) OR (((clinical*[Title]) AND guide*[Title]) AND manage*[Title])) OR ((best[Title]) AND practice*[Title])) OR ((evidence[Title]) AND synthes*[Title])) OR ((consensus[Title]) AND develop*[Title])) OR ((practice[Title]) AND guideline*[Title])) OR (("evidence based"[Title]) AND guideline*[Title])) OR ((consensus[Title]) AND statement*[Title])) OR ((committee[Title]) AND opinion*[Title])) OR ((practice[Title]) AND bulletin*[Title])) OR ((clinical[Title]) AND recommendation*[Title])) OR ((("U.S. Preventive Services Task Force"[Title/Abstract]) OR USPSTF[Title/Abstract]) OR "United States Preventive Services Task Force"[Title/Abstract])) OR ACR Appropriateness Criteria[Title])) NOT ((((((("Letter"[Publication Type]) OR "News"[Publication Type]) OR "Patient Education Handout"[Publication Type]) OR "Comment"[Publication Type]) OR "Editorial"[Publication Type])) OR "Newspaper Article"[Publication Type]))) NOT (("Animals"[Mesh]) NOT (("Animals"[Mesh]) AND "Humans"[Mesh])))))
Acknowledgements
We thank David R. Goldmann, MD for his contribution to the development of the clinical question and review protocol. This work is supported by Elsevier Evidence-based Medicine Center.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Chamberlain JJ, Rhinehart AS, Shaefer CF Jr, et al. Diagnosis and Management of Diabetes: Synopsis of the 2016 American Diabetes Association Standards of Medical Care in Diabetes. Ann Intern Med 2016;164:542-52. [Crossref] [PubMed]
- Fox CS, Golden SH, Anderson C, et al. Update on Prevention of Cardiovascular Disease in Adults With Type 2 Diabetes Mellitus in Light of Recent Evidence: A Scientific Statement From the American Heart Association and the American Diabetes Association. Diabetes Care 2015;38:1777-803. [Crossref] [PubMed]
- Solini A, Grossman E. What Should Be the Target Blood Pressure in Elderly Patients With Diabetes? Diabetes Care 2016;39 Suppl 2:S234-43. [Crossref] [PubMed]
- Tankeu AT, Noubiap JJ. Oscillating blood pressure therapeutic target in type 2 diabetes patients with hypertension. Ann Transl Med 2016;4:422. [Crossref] [PubMed]
- Brunström M, Eliasson M, Nilsson PM, et al. Blood pressure treatment levels and choice of antihypertensive agent in people with diabetes mellitus: an overview of systematic reviews. J Hypertens 2017;35:453-62. [Crossref] [PubMed]
- Higgins J, Green S. Cochrane handbook for systematic reviews of interventions. Version 5.1.0. Cochrane book series. London: The Cochrane Collaboration, 2011.
- Slutsky J, Atkins D, Chang S, et al. AHRQ series paper 1: comparing medical interventions: AHRQ and the effective health-care program. J Clin Epidemiol 2010;63:481-3. [Crossref] [PubMed]
- Hartling L, Guise JM, Kato E, et al. EPC Methods: An Exploration of Methods and Context for the Production of Rapid Reviews AHRQ Comparative Effectiveness Reviews 2015. [Internet]. [PubMed]
- Robinson KA, Chou R, Berkman ND, et al. Integrating Bodies of Evidence: Existing Systematic Reviews and Primary Studies. Methods Guide for Effectiveness and Comparative Effectiveness Reviews. AHRQ Methods for Effective Health Care. Rockville 2008.
- Fu R, Gartlehner G, Grant M, et al. Conducting quantitative synthesis when comparing medical interventions: AHRQ and the Effective Health Care Program. J Clin Epidemiol 2011;64:1187-97. [Crossref] [PubMed]
- Palmer TM, Sterne JA. Meta-Analysis in Stata: An Updated Collection from the Stata Journal. Second Edition. Texas: Stata Press, 2016.
- Treadwell JR, Uhl S, Tipton K, et al. Assessing equivalence and noninferiority. J Clin Epidemiol 2012;65:1144-9. [Crossref] [PubMed]
- Yuan KH, Maxwell S. On the post hoc power in testing mean differences. J Educ Behav Stat 2005;30:141-67. [Crossref]
- Goodman SN, Berlin JA. The use of predicted confidence intervals when planning experiments and the misuse of power when interpreting results. Ann Intern Med 1994;121:200-6. [Crossref] [PubMed]
- Levine M, Ensom MH. Post hoc power analysis: an idea whose time has passed? Pharmacotherapy 2001;21:405-9. [Crossref] [PubMed]
- Guyatt GH, Oxman AD, Kunz R, et al. GRADE guidelines 6. Rating the quality of evidence--imprecision. J Clin Epidemiol 2011;64:1283-93. [Crossref] [PubMed]
- Balshem HS, Ansari M, Norris S, et al. Finding Grey Literature Evidence and Assessing for Outcome and Analysis Reporting Biases When Comparing Medical Interventions: AHRQ and the Effective Health Care Program. Available online: www.effectivehealthcare.ahrq.gov/reports/final.cfm
- Sterne JA, Sutton AJ, Ioannidis JP, et al. Recommendations for examining and interpreting funnel plot asymmetry in meta-analyses of randomised controlled trials. BMJ 2011;343:d4002. [Crossref] [PubMed]
- Guyatt GH, Oxman AD, Vist G, et al. GRADE guidelines: 4. Rating the quality of evidence--study limitations (risk of bias). J Clin Epidemiol 2011;64:407-15. [Crossref] [PubMed]
- Guyatt GH, Oxman AD, Montori V, et al. GRADE guidelines: 5. Rating the quality of evidence--publication bias. J Clin Epidemiol 2011;64:1277-82. [Crossref] [PubMed]
- Guyatt GH, Oxman AD, Kunz R, et al. GRADE guidelines: 8. Rating the quality of evidence--indirectness. J Clin Epidemiol 2011;64:1303-10. [Crossref] [PubMed]
- Andrews J, Guyatt G, Oxman AD, et al. GRADE guidelines: 14. Going from evidence to recommendations: the significance and presentation of recommendations. J Clin Epidemiol 2013;66:719-25. [Crossref] [PubMed]
- Grading of Recommendations Assessment DaEGWG. Available online: http://gdt.guidelinedevelopment.org/central_prod/_design/client/handbook/handbook.html#h.fueh5iz0cor4
- Cushman WC, Evans GW, Byington RP, et al. Effects of intensive blood-pressure control in type 2 diabetes mellitus. N Engl J Med 2010;362:1575-85. [Crossref] [PubMed]
- UK Prospective Diabetes Study Group. Tight blood pressure control and risk of macrovascular and microvascular complications in type 2 diabetes: UKPDS 38. BMJ 1998;317:703-13. [Crossref] [PubMed]
- Estacio RO, Coll JR, Tran ZV, et al. Effect of intensive blood pressure control with valsartan on urinary albumin excretion in normotensive patients with type 2 diabetes. Am J Hypertens 2006;19:1241-8. [Crossref] [PubMed]
- Schrier RW, Estacio RO, Esler A, et al. Effects of aggressive blood pressure control in normotensive type 2 diabetic patients on albuminuria, retinopathy and strokes. Kidney Int 2002;61:1086-97. [Crossref] [PubMed]
- Estacio RO, Jeffers BW, Gifford N, et al. Effect of blood pressure control on diabetic microvascular complications in patients with hypertension and type 2 diabetes. Diabetes Care 2000;23:B54-64. [PubMed]
- Hansson L, Zanchetti A, Carruthers SG, et al. Effects of intensive blood-pressure lowering and low-dose aspirin in patients with hypertension: principal results of the Hypertension Optimal Treatment (HOT) randomised trial. HOT Study Group. Lancet 1998;351:1755-62. [Crossref] [PubMed]
- Xie X, Atkins E, Lv J, et al. Intensive blood pressure lowering–Authors' reply. Lancet 2016;387:2291. [Crossref] [PubMed]
- Brunström M, Carlberg B. Effect of antihypertensive treatment at different blood pressure levels in patients with diabetes mellitus: systematic review and meta-analyses. BMJ 2016;352:i717. [Crossref] [PubMed]
- Bundy JD, Li C, Stuchlik P, et al. Systolic Blood Pressure Reduction and Risk of Cardiovascular Disease and Mortality: A Systematic Review and Network Meta-analysis. JAMA Cardiol 2017;2:775-81. [Crossref] [PubMed]
- Ettehad D, Emdin CA, Kiran A, et al. Blood pressure lowering for prevention of cardiovascular disease and death: a systematic review and meta-analysis. Lancet 2016;387:957-67. [Crossref] [PubMed]
- Jerums G, Panagiotopoulos S, Ekinci E, et al. Cardiovascular outcomes with antihypertensive therapy in type 2 diabetes: an analysis of intervention trials. J Hum Hypertens 2015;29:473-7. [Crossref] [PubMed]
- Lv J, Ehteshami P, Sarnak MJ, et al. Effects of intensive blood pressure lowering on the progression of chronic kidney disease: a systematic review and meta-analysis. CMAJ 2013;185:949-57. [Crossref] [PubMed]
- McBrien K, Rabi DM, Campbell N, et al. Intensive and Standard Blood Pressure Targets in Patients With Type 2 Diabetes Mellitus: Systematic Review and Meta-analysis. Arch Intern Med 2012;172:1296-303. [Crossref] [PubMed]
- Thomopoulos C, Parati G, Zanchetti A. Effects of blood-pressure-lowering treatment on outcome incidence in hypertension. 11. Effects of total cardiovascular risk and achieved blood pressure: overview and meta-analyses of randomized trials. J Hypertens 2017;35:2138-49. [Crossref] [PubMed]
- Xie X, Atkins E, Lv J, et al. Effects of intensive blood pressure lowering on cardiovascular and renal outcomes: updated systematic review and meta-analysis. Lancet 2016;387:435-43. [Crossref] [PubMed]
- Feldstein CA. Lowering blood pressure to prevent stroke recurrence: a systematic review of long-term randomized trials. J Am Soc Hypertens 2014;8:503-13. [Crossref] [PubMed]
- Arguedas JA, Leiva V, Wright JM. Blood pressure targets for hypertension in people with diabetes mellitus. Cochrane Database Syst Rev 2013;10:CD008277. [PubMed]
- Estacio RO, Schrier RW. Antihypertensive therapy in type 2 diabetes: implications of the appropriate blood pressure control in diabetes (ABCD) trial. Am J Cardiol 1998;82:9R-14R. [Crossref] [PubMed]
- Buckley LF, Dixon DL, Wohlford GF 4th, et al. Effect of Intensive Blood Pressure Control in Patients with Type 2 Diabetes Mellitus Over 9 Years of Follow-Up: A Subgroup Analysis of High-Risk ACCORDION Trial Participants. Diabetes Obes Metab 2018;20:1499-502. [Crossref] [PubMed]
- Soliman EZ, Byington RP, Bigger JT, et al. Effect of Intensive Blood Pressure Lowering on Left Ventricular Hypertrophy in Patients With Diabetes Mellitus: Action to Control Cardiovascular Risk in Diabetes Blood Pressure Trial. Hypertension 2015;66:1123-9. [PubMed]
- Margolis KL, Palermo L, Vittinghoff E, et al. Intensive blood pressure control, falls, and fractures in patients with type 2 diabetes: the ACCORD trial. J Gen Intern Med 2014;29:1599-606. [Crossref] [PubMed]
- Pigott T, Noyes J, Umscheid CA, et al. AHRQ series on complex intervention systematic reviews-paper 5: advanced analytic methods. J Clin Epidemiol 2017;90:37-42. [Crossref] [PubMed]
- Berlowitz DR, Foy CG, Kazis LE, et al. Effect of Intensive Blood-Pressure Treatment on Patient-Reported Outcomes. N Engl J Med 2017;377:733-44. [Crossref] [PubMed]
- Jones AP, Riley RD, Williamson PR, et al. Meta-analysis of individual patient data versus aggregate data from longitudinal clinical trials. Clin Trials 2009;6:16-27. [Crossref] [PubMed]
- Wu JS, Yang YC, Lu FH, et al. Population-based study on the prevalence and risk factors of orthostatic hypotension in subjects with pre-diabetes and diabetes. Diabetes Care 2009;32:69-74. [Crossref] [PubMed]
- Wolters FJ, Mattace-Raso FU, Koudstaal PJ, et al. Orthostatic Hypotension and the Long-Term Risk of Dementia: A Population-Based Study. PLoS Med 2016;13:e1002143. [Crossref] [PubMed]
- Bakris G, Sorrentino M. Redefining Hypertension - Assessing the New Blood-Pressure Guidelines. N Engl J Med 2018;378:497-9. [Crossref] [PubMed]
- National Institute for Health and Care Excellence. Type 2 diabetes in adults: management. NICE guidelines 2015.
- Rosendorff C, Lackland DT, Allison M, et al. Treatment of hypertension in patients with coronary artery disease: A scientific statement from the American Heart Association, American College of Cardiology, and American Society of Hypertension. Circulation 2015;131:e435-70.
- The Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure [Internet] 2016.
- Standards of medical care in diabetes-2016: Summary of revisions. Diabetes Care 2016;39:S4-5. [Crossref] [PubMed]
- de Boer IH, Bangalore S, Benetos A, et al. Diabetes and Hypertension: A Position Statement by the American Diabetes Association. Diabetes Care 2017;40:1273-84. [Crossref] [PubMed]
- Whelton PK, Carey RM, Aronow WS, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol 2018;71:e127-248. [Crossref] [PubMed]
- American Diabetes Association. 1. Improving Care and Promoting Health in Populations: Standards of Medical Care in Diabetes-2018. Diabetes Care 2018;41:S7-12. [Crossref] [PubMed]
- American Diabetes Association. 4. Lifestyle Management: Standards of Medical Care in Diabetes-2018. Diabetes Care 2018;41:S38-50. [Crossref] [PubMed]
- Furie KL, Kasner SE, Adams RJ, et al. Guidelines for the prevention of stroke in patients with stroke or transient ischemic attack: a guideline for healthcare professionals from the american heart association/american stroke association. Stroke 2011;42:227-76. [Crossref] [PubMed]
- American Diabetes Association. Standards of Medical Care in Diabetes - 2012. Diabetes Care 2012;35:S11-63. [Crossref] [PubMed]
- Summary of Revisions: Standards of Medical Care in Diabetes-2018. Diabetes Care 2018;41:S4-6. [Crossref] [PubMed]
- Network SI. Management of diabetes: a national clinical guideline, 2010.
- Krause T, Lovibond K, Caulfield M, et al. Management of hypertension: Summary of NICE guidance. BMJ 2011;343:d4891. [Crossref] [PubMed]
- Houle SK, Padwal R, Tsuyuki RT. The 2012-2013 Canadian Hypertension Education Program (CHEP) guidelines for pharmacists: An update. Can Pharm J (Ott) 2013;146:146-50. [Crossref] [PubMed]
- Task Force for the management of arterial hypertension of the European Society of Hypertension; Task Force for the management of arterial hypertension of the European Society of Cardiology. 2013 ESH/ESC guidelines for the management of arterial hypertension. Blood Pressure 2013;22:193-278. [Crossref] [PubMed]
- Moreno G, Mangione CM, Kimbro L, et al. Guidelines abstracted from the American Geriatrics Society guidelines for improving the care of older adults with diabetes mellitus: 2013 update. J Am Geriatr Soc 2013;61:2020-6. [Crossref] [PubMed]
- Rodriguez-Gutierrez R, Ospina NS, McCoy RG, et al. Inclusion of hypoglycemia in clinical practice guidelines and performance measures in the care of patients with diabetes. JAMA Intern Med 2016;176:1714-6. [PubMed]
- Rodriguez-Gutierrez R, Lipska KJ, McCoy RG, et al. Hypoglycemia as an indicator of good diabetes care. BMJ 2016;352:i1084. [Crossref] [PubMed]
- American Diabetes Association. 6. Glycemic Targets: Standards of Medical Care in Diabetes-2018. Diabetes Care 2018;41:S55-64. [Crossref] [PubMed]
- Mills KT, Obst KM, Shen W, et al. Comparative Effectiveness of Implementation Strategies for Blood Pressure Control in Hypertensive Patients: A Systematic Review and Meta-analysis. Ann Intern Med 2018;168:110-20. [Crossref] [PubMed]
- Kahya Eren N, Harman E, Dolek D, et al. Rate of blood pressure control and antihypertensive treatment approaches in diabetic patients with hypertension. Turk Kardiyol Dern Ars 2014;42:733-40. [Crossref] [PubMed]
- Schmieder RE, Gitt AK, Koch C, et al. Achievement of individualized treatment targets in patients with comorbid type-2 diabetes and hypertension: 6 months results of the DIALOGUE registry. BMC Endocr Disord 2015;15:23. [Crossref] [PubMed]
- Wang J, Shi X, Ma C, et al. Visit-to-visit blood pressure variability is a risk factor for all-cause mortality and cardiovascular disease: a systematic review and meta-analysis. J Hypertens 2017;35:10-7. [Crossref] [PubMed]
- Karmali KN, Persell SD, Perel P, et al. Risk scoring for the primary prevention of cardiovascular disease. Cochrane Database Syst Rev 2017;3:CD006887. [PubMed]
- Stevens SL, Wood S, Koshiaris C, et al. Blood pressure variability and cardiovascular disease: systematic review and meta-analysis. BMJ 2016;354:i4098. [Crossref] [PubMed]
- Diaz KM, Tanner RM, Falzon L, et al. Visit-to-visit variability of blood pressure and cardiovascular disease and all-cause mortality: a systematic review and meta-analysis. Hypertension 2014;64:965-82. [Crossref] [PubMed]
- Kollias A, Ntineri A, Stergiou GS. Association of night-time home blood pressure with night-time ambulatory blood pressure and target-organ damage: a systematic review and meta-analysis. J Hypertens 2017;35:442-52. [Crossref] [PubMed]
- Salles GF, Reboldi G, Fagard RH, et al. Prognostic Effect of the Nocturnal Blood Pressure Fall in Hypertensive Patients: The Ambulatory Blood Pressure Collaboration in Patients With Hypertension (ABC-H) Meta-Analysis. Hypertension 2016;67:693-700. [Crossref] [PubMed]
- Roush GC, Fagard RH, Salles GF, et al. Prognostic impact of sex-ambulatory blood pressure interactions in 10 cohorts of 17 312 patients diagnosed with hypertension: systematic review and meta-analysis. J Hypertens 2015;33:212-20. [Crossref] [PubMed]
- Xie JC, Yan H, Zhao YX, et al. Prognostic value of morning blood pressure surge in clinical events: a meta-analysis of longitudinal studies. J Stroke Cerebrovasc Dis 2015;24:362-9. [Crossref] [PubMed]
- Sheppard JP, Hodgkinson J, Riley R, et al. Prognostic significance of the morning blood pressure surge in clinical practice: a systematic review. Am J Hypertens 2015;28:30-41. [Crossref] [PubMed]
- Nomura K, Asayama K, Thijs L, et al. Thresholds for conventional and home blood pressure by sex and age in 5018 participants from 5 populations. Hypertension 2014;64:695-701. [Crossref] [PubMed]
- Brguljan-Hitij J, Thijs L, Li Y, et al. Risk stratification by ambulatory blood pressure monitoring across JNC classes of conventional blood pressure. Am J Hypertens 2014;27:956-65. [Crossref] [PubMed]
- Asayama K, Thijs L, Brguljan-Hitij J, et al. Risk stratification by self-measured home blood pressure across categories of conventional blood pressure: a participant-level meta-analysis. PLoS Med 2014;11:e1001591. [Crossref] [PubMed]
- Fuchs SC, Mello RG, Fuchs FC. Home blood pressure monitoring is better predictor of cardiovascular disease and target organ damage than office blood pressure: a systematic review and meta-analysis. Curr Cardiol Rep 2013;15:413. [Crossref] [PubMed]
- Omboni S, Gazzola T, Carabelli G, et al. Clinical usefulness and cost effectiveness of home blood pressure telemonitoring: meta-analysis of randomized controlled studies. J Hypertens 2013;31:455-67; discussion 67-8. [Crossref] [PubMed]
- American Diabetes Association. 9. Cardiovascular Disease and Risk Management: Standards of Medical Care in Diabetes-2018. Diabetes Care 2018;41:S86-104. [Crossref] [PubMed]
- Yannoutsos A, Kheder-Elfekih R, Halimi JM, et al. Should blood pressure goal be individualized in hypertensive patients? Pharmacol Res 2017;118:53-63. [Crossref] [PubMed]
- Wan EYF, Yu EYT, Fung CSC, et al. Do We Need a Patient-Centered Target for Systolic Blood Pressure in Hypertensive Patients With Type 2 Diabetes Mellitus? Hypertension 2017;70:1273-82. [Crossref] [PubMed]
- Brunström M, Carlberg B. Association of Blood Pressure Lowering With Mortality and Cardiovascular Disease Across Blood Pressure Levels: A Systematic Review and Meta-analysis. JAMA Intern Med 2018;178:28-36. [Crossref] [PubMed]
- Phillips RA, Xu J, Peterson LE, et al. Impact of Cardiovascular Risk on the Relative Benefit and Harm of Intensive Treatment of Hypertension. J Am Coll Cardiol 2018;71:1601-10. [Crossref] [PubMed]
- McManus RJ, Mant J, Franssen M, et al. Efficacy of self-monitored blood pressure, with or without telemonitoring, for titration of antihypertensive medication (TASMINH4): an unmasked randomised controlled trial. Lancet 2018;391:949-59. [Crossref] [PubMed]
- Emdin CA, Rahimi K, Neal B, et al. Blood pressure lowering in type 2 diabetes: a systematic review and meta-analysis. JAMA 2015;313:603-15. [Crossref] [PubMed]
- World Health Organization. A global brief on Hypertension. World Health Day 2013. Available online: http://ish-world.com/downloads/pdf/global_brief_hypertension.pdf
- James PA, Oparil S, Carter BL, et al. 2014 evidence-based guideline for the management of high blood pressure in adults: report from the panel members appointed to the Eighth Joint National Committee (JNC 8). JAMA 2014;311:507-20. [Crossref] [PubMed]
- Handelsman Y, Bloomgarden ZT, Grunberger G, et al. American association of clinical endocrinologists and american college of endocrinology - clinical practice guidelines for developing a diabetes mellitus comprehensive care plan - 2015. Endocr Pract 2015;21:1-87. [Crossref] [PubMed]
- Weber MA, Schiffrin EL, White WB, et al. Clinical Practice Guidelines for the Management of Hypertension in the Community: A Statement by the American Society of Hypertension and the International Society of Hypertension Clinical Practice Guidelines for the Management of Hypertension in the Community: A Statement by the American Society of Hypertension and the International Society of Hypertension. J Clin Hypertens (Greenwich) 2014;16:14-26. [Crossref] [PubMed]
- Chris Crawford. AAFP Decides to Not Endorse AHA/ACC Hypertension Guideline. Available online: https://wwwaafporg/patient-care/clinical-recommendations/non-endorsed.html
- Shea BJ, Hamel C, Wells GA, et al. AMSTAR is a reliable and valid measurement tool to assess the methodological quality of systematic reviews. J Clin Epidemiol 2009;62:1013-20. [Crossref] [PubMed]
- Viswanathan M, Berkman ND, Dryden DM, et al. Assessing Risk of Bias and Confounding in Observational Studies of Interventions or Exposures: Further Development of the RTI Item Bank. Rockville (MD): Agency for Healthcare Research and Quality (US); 2013.
- Higgins JP, Altman DG, Gotzsche PC, et al. The Cochrane Collaboration's tool for assessing risk of bias in randomised trials. BMJ 2011;343:d5928. [Crossref] [PubMed]
- Brouwers MC, Kho ME, Browman GP, et al. Development of the AGREE II, part 2: assessment of validity of items and tools to support application. CMAJ 2010;182:E472-8. [Crossref] [PubMed]
- Brouwers MC, Kho ME, Browman GP, et al. Development of the AGREE II, part 1: performance, usefulness and areas for improvement. CMAJ 2010;182:1045-52. [Crossref] [PubMed]
- Ó Hartaigh B. Szymonifka J, Okin PM. Achieving target SBP for lowering the risk of major adverse cardiovascular events in persons with diabetes mellitus. J Hypertens 2018;36:101-9. [Crossref] [PubMed]
- Barzilay JI, Howard AG, Evans GW, et al. Intensive blood pressure treatment does not improve cardiovascular outcomes in centrally obese hypertensive individuals with diabetes: the Action to Control Cardiovascular Risk in Diabetes (ACCORD) Blood Pressure Trial. Diabetes Care 2012;35:1401-5. [Crossref] [PubMed]
- Chen LY, Bigger JT, Hickey KT, et al. Effect of Intensive Blood Pressure Lowering on Incident Atrial Fibrillation and P-Wave Indices in the ACCORD Blood Pressure Trial. Am J Hypertens 2016;29:1276-82. [Crossref] [PubMed]
- Margolis KL, O'Connor PJ, Morgan TM, et al. Outcomes of combined cardiovascular risk factor management strategies in type 2 diabetes: the ACCORD randomized trial. Diabetes Care 2014;37:1721-8. [Crossref] [PubMed]
- The Hypertension Detection and Follow-up Program Cooperative Research Group. Mortality findings for stepped-care and referred-care participants in the hypertension detection and follow-up program, stratified by other risk factors. Prev Med 1985;14:312-35. [Crossref] [PubMed]
- Wright RA, Judson FN. Five-year findings of the hypertension detection and follow-up program. JAMA 1997;277:157-66. [Crossref] [PubMed]
- Gundersen T, Kjekshus J. Timolol treatment after myocardial infarction in diabetic patients. Diabetes Care 1983;6:285-90. [Crossref] [PubMed]
- Hypertension-Stroke Cooperative Study Group. Effect of Antihypertensive Treatment on Stroke Recurrence. JAMA 1974;229:409-18. [Crossref] [PubMed]
- Hypertension Detection and Follow-up Program Cooperative Group. Five-Year Findings of the Hypertension Detection and Follow-up Program: I. Reduction in Mortality of Persons With High Blood Pressure, Including Mild Hypertension. JAMA 1979;242:2562-71. [Crossref] [PubMed]
- Amery A, Brixko P, Clement D, et al. Mortality and morbidity results from the european working party on high blood pressure in the elderly trial. Lancet 1985;1:1349-54. [Crossref] [PubMed]
- Langford HG, Stamier J, Wassertheil-Smoller S, et al. All-cause mortality in the hypertension detection and follow-up program: Findings for the whole cohort and for persons with less severe hypertension, with and without other traits related to risk of mortality. Prog Cardiovasc Dis 1986;29:29-54. [Crossref] [PubMed]
- Gustafsson I, Torp-Pedersen C, Køber L, et al. Effect of the angiotensin-converting enzyme inhibitor trandolapril on mortality and morbidity in diabetic patients with left ventricular dysfunction after acute myocardial infarction. J Am Coll Cardiol 1999;34:83-9. [Crossref] [PubMed]
- Yusuf S, Pitt B, Davis CE, et al. Effect of Enalapril on Mortality and the Development of Heart Failure in Asymptomatic Patients with Reduced Left Ventricular Ejection Fractions. N Engl J Med 1992;327:685-91. [Crossref] [PubMed]
- Yusuf S, Pitt B, Davis CE, et al. Effect of enalapril on survival in patients with reduced left ventricular ejection fractions and congestive heart failure. N Engl J Med 1991;325:293-302. [Crossref] [PubMed]
- Moyé LA, Pfeffer MA, Wun CC, et al. Uniformity of captopril benefit in the SAVE study: Subgroup analysis. Survival and Ventricular Enlargement Study. Eur Heart J 1994;15:2-8. [Crossref] [PubMed]
- Hjalmarson A, Goldstein S, Fagerberg B, et al. Effect of metoprolol CR/XL in chronic heart failure: Metoprolol CR/XL Randomised Intervention Trial in Congestive Heart Failure (MERIT-HF). Lancet 1999;353:2001-7. [Crossref] [PubMed]
- Swedberg K, Held P, Kjekshus J, et al. Effects of the Early Administration of Enalapril on Mortality in Patients with Acute Myocardial Infarction: Results of the Cooperative New Scandinavian Enalapril Survival Study II (Consensus II). N Engl J Med 1992;327:678-84. [Crossref] [PubMed]
- Dahlöf B, Hansson L, Lindholm LH, et al. Morbidity and mortality in the Swedish Trial in Old Patients with Hypertension (STOP-Hypertension). Lancet 1991;338:1281-5. [Crossref] [PubMed]
- SHEP Cooperative Research Group. Prevention of Stroke by Antihypertensive Drug Treatment in Older Persons With Isolated Systolic Hypertension: Final Results of the Systolic Hypertension in the Elderly Program (SHEP). JAMA 1991;265:3255-64. [Crossref] [PubMed]
- Pfeffer MA, Braunwald E, Moyé LA, et al. Effect of Captopril on Mortality and Morbidity in Patients with Left Ventricular Dysfunction after Myocardial Infarction: Results of the Survival and Ventricular Enlargement Trial. N Engl J Med 1992;327:669-77. [Crossref] [PubMed]
- Lewis EJ, Hunsicker LG, Bain RP, et al. The Effect of Angiotensin-Converting-Enzyme Inhibition on Diabetic Nephropathy. N Engl J Med 1993;329:1456-62. [Crossref] [PubMed]
- Køber L, Torp-Pedersen C, Carlsen JE, et al. A clinical trial of the angiotensin-converting–enzyme inhibitor trandolapril in patients with left ventricular dysfunction after myocardial infarction. N Engl J Med 1995;333:1670-6. [Crossref] [PubMed]
- Laffel LM, McGill JB, Gans DJ. The beneficial effect of angiotensin-converting enzyme inhibition with captopril on diabetic nephropathy in normotensive IDDM patients with microalbuminuria. Am J Med 1995;99:497-504. [Crossref] [PubMed]
- Curb JD, Pressel SL, Cutler JA, et al. Effect of diuretic-based antihypertensive treatment on cardiovascular disease risk in older diabetic patients with isolated systolic hypertension. JAMA 1996;276:1886-92. [Crossref] [PubMed]
- Shindler DM, Kostis JB, Yusuf S, et al. Diabetes mellitus, a predictor of morbidity and mortality in the Studies of Left Ventricular Dysfunction (SOLVD) trials and registry. Am J Cardiol 1996;77:1017-20. [Crossref] [PubMed]
- Staessen JA, Fagard R, Thijs L, et al. Randomised double-blind comparison of placebo and active treatment for older patients with isolated systolic hypertension. Lancet 1997;350:757-64. [Crossref] [PubMed]
- UK Prospective Diabetes Study Group. Efficacy of atenolol and captopril in reducing risk of macrovascular and microvascular complications in type 2 diabetes: UKPDS 39. BMJ 1998;317:713-20. [Crossref] [PubMed]
- Estacio RO, Jeffers BW, Hiatt WR, et al. The effect of nisoldipine as compared with enalapril on cardiovascular outcomes in patients with non-insulin-dependent diabetes and hypertension. N Engl J Med 1998;338:645-52. [Crossref] [PubMed]
- Holman R, Turner R, Stratton I, et al. Efficacy of atenolol and captopril in reducing risk of macrovascular and microvascular complications in type 2 diabetes: UKPDS 39. BMJ 1998;317:713-20. [Crossref] [PubMed]
- Liu L, Wang JG, Gong L, et al. Comparison of active treatment and placebo in older Chinese patients with isolated systolic hypertension. J Hypertens 1998;16:1823-9. [Crossref] [PubMed]
- Ravid M, Brosh D, Levi Z, et al. Use of enalapril to attenuate decline in renal function in normotensive, normoalbuminuric patients with type 2 diabetes mellitus: A randomized, controlled trial. Ann Intern Med 1998;128:982-8. [Crossref] [PubMed]
- Tuomilehto J, Rastenyte D, Birkenhager WH, et al. Effects of calcium-channel blockade in older patients with diabetes and systolic hypertension. N Engl J Med 1999;340:677-84. [Crossref] [PubMed]
- Schrier RW, Estacio RO. Additional follow-up from the ABCD trial in patients with type 2 diabetes and hypertension. N Engl J Med 2000;343:1969. [Crossref] [PubMed]
- Lindholm LH, Hansson L, Ekbom T, et al. Comparison of antihypertensive treatments in preventing cardiovascular events in elderly diabetic patients: Results from the Swedish trial in old patients with hypertension - 2. J Hypertens 2000;18:1671-5. [Crossref] [PubMed]
- Hansson L, Hedner T, Lund-Johansen P, et al. Randomised trial of effects of calcium antagonists compared with diuretics and β-blockers on cardiovascular morbidity and mortality in hypertension: The Nordic Diltiazem (NORDIL) study. Lancet 2000;356:359-65. [Crossref] [PubMed]
- Lindholm LH, Ibsen H, Dahlöf B, et al. Cardiovascular morbidity and mortality in patients with diabetes in the Losartan Intervention For Endpoint reduction in hypertension study (LIFE): A randomised trial against atenolol. Lancet 2002;359:1004-10. [Crossref] [PubMed]
- Niskanen L, Hedner T, Hansson L, et al. Reduced cardiovascular morbidity and mortality in hypertensive diabetic patients on first-line therapy with an ACE inhibitor compared with a diuretic/β-blocker-based treatment regimen: A subanalysis of the captopril prevention project. Diabetes Care 2001;24:2091-6. [Crossref] [PubMed]
- Erdmann E, Philippe L, Patricia V, et al. Results from post-hoc analyses of the CIBIS II trial: Effect of bisoprolol in high-risk patient groups with chronic heart failure. Eur J Heart Fail 2001;3:469-79. [Crossref] [PubMed]
- Major outcomes in high-risk hypertensive patients randomized to angiotensin-converting enzyme inhibitor or calcium channel blocker vs diuretic: The antihypertensive and lipid-lowering treatment to prevent heart attack trial (ALLHAT). JAMA 2002;288:2981-97. [Crossref] [PubMed]
- Effects of ramipril on cardiovascular and microvascular outcomes in people with diabetes mellitus: results of the HOPE study and MICRO-HOPE substudy. Heart Outcomes Prevention Evaluation Study Investigators. Lancet 2000;355:253-9. [Crossref] [PubMed]
- Gerstein HC, Yusuf S, Mann JFE, et al. Effects of ramipril on cardiovascular and microvascular outcomes in people with diabetes mellitus: Results of the HOPE study and MICRO-HOPE substudy. Lancet 2000;355:253-9. [Crossref] [PubMed]
- O'Hare P, Bilous R, Mitchell T, et al. Low-dose ramipril reduces microalbuminuria in type 1 diabetic patients without hypertension: Results of a randomized controlled trial. Diabetes Care 2000;23:1823-9. [Crossref] [PubMed]
- Wang JG, Staessen JA, Gong L, et al. Chinese trial on isolated systolic hypertension in the elderly. Systolic Hypertension in China (Syst-China) Collaborative Group. Arch Intern Med 2000;160:211-20. [Crossref] [PubMed]
- Brenner BM, Cooper ME, De Zeeuw D, et al. Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N Engl J Med 2001;345:861-9. [Crossref] [PubMed]
- Cohn JN, Tognoni G. A randomized trial of the angiotensin-receptor blocker valsartan in chronic heart failure. N Engl J Med 2001;345:1667-75. [Crossref] [PubMed]
- Lewis EJ, Hunsicker LG, Clarke WR, et al. Renoprotective effect of the angiotensin-receptor antagonist irbesartan in patients with nephropathy due to type 2 diabetes. N Engl J Med 2001;345:851-60. [Crossref] [PubMed]
- MacMahon S, Neal B, Tzourio C, et al. Randomised trial of a perindopril-based blood-pressure-lowering regimen among 6105 individuals with previous stroke or transient ischaemic attack. Lancet 2001;358:1033-41. [Crossref] [PubMed]
- Parving HH, Lehnert H, Brochner-Mortensen J, et al. The effect of irbesartan on the development of diabetic nephropathy in patients with type 2 diabetes. N Engl J Med 2001;345:870-8. [Crossref] [PubMed]
- Fogari R, Preti P, Zoppi A, et al. Effects of amlodipine fosinopril combination on microalbuminuria in hypertensive type 2 diabetic patients. Am J Hypertens 2002;15:1042-9. [Crossref] [PubMed]
- Schrader J, Lüders S, Kulschewski A, et al. Morbidity and mortality after stroke, eprosartan compared with nitrendipine for secondary prevention: Principal results of a prospective randomized controlled study (MOSES). Stroke 2005;36:1218-26. [Crossref] [PubMed]
- Yui Y, Sumiyoshi T, Kodama K, et al. Nifedipine retard was an effective as angiotensin converting enzyme inhibitors in preventing cardiac events in high-risk hypertensive patients with diabetes and coronary artery disease: The Japan Multicenter Investigation for Cardiovascular Diseases-B (JMIC-B) subgroup analysis. Hypertens Res 2004;27:449-56. [Crossref] [PubMed]
- Bakris GL, Gaxiola E, Messerli FH, et al. Clinical outcomes in the diabetes cohort of the international verapamil SR-trandolapril study. Hypertension 2004;44:637-42. [Crossref] [PubMed]
- Mancia G, Brown M, Castaigne A, et al. Outcomes with nifedipine GITS or Co-amilozide in hypertensive diabetics and nondiabetics in intervention as a goal in hypertension (INSIGHT). Hypertension 2003;41:431-6. [Crossref] [PubMed]
- Domanski M, Krause-Steinrauf H, Deedwania P, et al. The effect of diabetes on outcomes of patients with advanced heart failure in the BEST trial. J Am Coll Cardiol 2003;42:914-22. [Crossref] [PubMed]
- Berl T, Hunsicker LG, Lewis JB, et al. Cardiovascular outcomes in the Irbesartan Diabetic Nephropathy Trial of patients with type 2 diabetes and overt nephropathy. Ann Intern Med 2003;138:542-9. [Crossref] [PubMed]
- Dens JA, Desmet WJ, Coussement P, et al. Long term effects of nisoldipine on the progression of coronary atherosclerosis and the occurrence of clinical events: The NICOLE study. Heart 2003;89:887-92. [Crossref] [PubMed]
- Fox KM, Bertrand M, Ferrari R, et al. Efficacy of perindopril in reduction of cardiovascular events among patients with stable coronary artery disease: Randomised, double-blind, placebo-controlled, multicentre trial (the EUROPA study). Lancet 2003;362:782-8. [Crossref] [PubMed]
- Havranek EP, Esler A, Estacio RO, et al. Differential effects of antihypertensive agents on electrocardiographic voltage: results from the Appropriate Blood Pressure Control in Diabetes (ABCD) trial. Am Heart J 2003;145:993-8. [Crossref] [PubMed]
- Lithell H, Hansson L, Skoog I, et al. The study on cognition and prognosis in the elderly (SCOPE): Principal results of a randomized double-blind intervention trial. J Hypertens 2003;21:875-86. [Crossref] [PubMed]
- Mehler PS, Coll JR, Estacio R, et al. Intensive blood pressure control reduces the risk of cardiovascular events in patients with peripheral arterial disease and type 2 diabetes. Circulation 2003;107:753-6. [Crossref] [PubMed]
- Berthet K, Neal BC, Chalmers JP, et al. Reductions in the risks of recurrent stroke in patients with and without diabetes: the PROGRESS Trial. Blood Press 2004;13:7-13. [Crossref] [PubMed]
- Braunwald E, Domanski MJ, Fowler SE, et al. Angiotensin-converting-enzyme inhibition in stable coronary artery disease. N Engl J Med 2004;351:2058-68. [Crossref] [PubMed]
- Marre M, Lievre M, Chatellier G, et al. Effects of low dose ramipril on cardiovascular and renal outcomes in patients with type 2 diabetes and raised excretion of urinary albumin: Randomised, double blind, placebo controlled trial (the DIABHYCAR study). BMJ 2004;328:495-9. [Crossref] [PubMed]
- Nissen SE, Tuzcu EM, Libby P, et al. Effect of antihypertensive agents on cardiovascular events in patients with coronary disease and normal blood pressure. The CAMELOT study: A randomized controlled trial. JAMA 2004;292:2217-25. [Crossref] [PubMed]
- Poole-Wilson PA, Lubsen PJ, Kirwan BA, et al. Effect of long-acting nifedipine on mortality and cardiovascular morbidity in patients with stable angina requiring treatment (ACTION trial): Randomised controlled trial. Lancet 2004;364:849-57. [Crossref] [PubMed]
- Ruggenenti P, Fassi A, Ilieva AP, et al. Preventing microalbuminuria in type 2 diabetes. N Engl J Med 2004;351:1941-51. [Crossref] [PubMed]
- Daly CA, Fox KM, Remme WJ, et al. The effect of perindopril on cardiovascular morbidity and mortality in patients with diabetes in the EUROPA study: Results from the PERSUADE substudy. Eur Heart J 2005;26:1369-78. [Crossref] [PubMed]
- Deedwania PC, Giles TD, Klibaner M, et al. Efficacy, safety and tolerability of metoprolol CR/XL in patients with diabetes and chronic heart failure: Experiences from MERIT-HF. Am Heart J 2005;149:159-67. [Crossref] [PubMed]
- Kowey PR, Dickson TZ, Zhang Z, et al. Losartan and end-organ protection - Lessons from the RENAAL study. Clin Cardiol 2005;28:136-42. [Crossref] [PubMed]
- Liu L, Zhang Y, Liu G, et al. The Felodipine Event Reduction (FEVER) Study: A randomized long-term placebo-controlled trial in Chinese hypertensive patients. J Hypertens 2005;23:2157-72. [Crossref] [PubMed]
- Trenkwalder P, Elmfeldt D, Hofman A, et al. The study on Cognition and Prognosis in the Elderly (SCOPE) - Major CV events and stroke in subgroups of patients. Blood Pressure 2005;14:31-7. [Crossref] [PubMed]
- Schrader J, Hammersen F, Lüders S, et al. Morbidity and mortality after stroke in patients with diabetes - Subgroup analysis from the MOSES study. Journal of Clinical and Basic Cardiology 2006;9:2-5.
- Bilous R, Chaturvedi N, Sjølie AK, et al. Effect of Candesartan on Microalbuminuria and Albumin Excretion Rate in Diabetes Three Randomized Trials. Ann Intern Med 2009;151:11-20. [Crossref] [PubMed]
- Östergren J, Poulter NR, Sever PS, et al. The Anglo-Scandinavian Cardiac Outcomes Trial: Blood pressure-lowering limb: Effects in patients with type II diabetes. J Hypertens 2008;26:2103-11. [Crossref] [PubMed]
- McLean RC, Mohler ER, Blumenthal RS. Vascular Disease, Hypertension, and Prevention. J Am Coll Cardiol 2006;47:D3-8. [Crossref] [PubMed]
- Schrier RW, Estacio RO, Mehler PS, et al. Appropriate blood pressure control in hypertensive and normotensive type 2 diabetes mellitus: a summary of the ABCD trial. Nat Clin Pract Nephrol 2007;3:428-38. [Crossref] [PubMed]
- Wagener G. Calcium antagonists and hypertension: Role of co-existent coronary disease impaired renal function and diabetes. Rotterdam: Erasmus University Rotterdam, 2007.
- Yusuf S, Teo K, Anderson C, et al. Effects of the angiotensin-receptor blocker telmisartan on cardiovascular events in high-risk patients intolerant to angiotensin-converting enzyme inhibitors: a randomised controlled trial. Lancet 2008;372:1174-83. [Crossref] [PubMed]
- JATOS Study Group. Principal results of the Japanese trial to assess optimal systolic blood pressure in elderly hypertensive patients (JATOS). Hypertens Res 2008;31:2115-27. [Crossref] [PubMed]
- Chaturvedi N, Porta M, Klein R, et al. Effect of candesartan on prevention (DIRECT-Prevent 1) and progression (DIRECT-Protect 1) of retinopathy in type 1 diabetes: randomised, placebo-controlled trials. Lancet 2008;372:1394-402. [Crossref] [PubMed]
- Howard BV, Roman MJ, Devereux RB, et al. Effect of lower targets for blood pressure and LDL cholesterol on atherosclerosis in diabetes: The SANDS randomized trial. JAMA 2008;299:1678-89. [Crossref] [PubMed]
- Lüders S, Schrader J, Berger J, et al. The PHARAO study: Prevention of hypertension with the angiotensin- converting enzyme inhibitor ramipril in patients with high-normal blood pressure - A prospective, randomized, controlled prevention trial of the German Hypertension League. J Hypertens 2008;26:1487-96. [Crossref] [PubMed]
- Massie BM, Carson PE, McMurray JJ, et al. Irbesartan in patients with heart failure and preserved ejection fraction. N Engl J Med 2008;359:2456-67. [Crossref] [PubMed]
- Sjølie AK, Klein R, Porta M, et al. Effect of candesartan on progression and regression of retinopathy in type 2 diabetes (DIRECT-Protect 2): a randomised placebo-controlled trial. Lancet 2008;372:1385-93. [Crossref] [PubMed]
- Yusuf S, Diener HC, Sacco RL, et al. Telmisartan to prevent recurrent stroke and cardiovascular events. N Engl J Med 2008;359:1225-37. [Crossref] [PubMed]
- de Galan BE, Perkovic V, Ninomiya T, et al. Lowering blood pressure reduces renal events in type 2 diabetes. J Am Soc Nephrol 2009;20:883-92. [Crossref] [PubMed]
- Mauer M, Zinman B, Gardiner R, et al. Renal and retinal effects of enalapril and losartan in type 1 diabetes. N Engl J Med 2009;361:40-51. [Crossref] [PubMed]
- Nakao K, Hirata M, Oba K, et al. Role of diabetes and obesity in outcomes of the candesartan antihypertensive survival evaluation in Japan (CASE-J) trial. Hypertens Res 2010;33:600-6. [Crossref] [PubMed]
- Weber MA, Bakris GL, Jamerson K, et al. Cardiovascular Events During Differing Hypertension Therapies in Patients With Diabetes. J Am Coll Cardiol 2010;56:77-85. [Crossref] [PubMed]
- Chew EY, Ambrosius WT, Davis MD, et al. Effects of medical therapies on retinopathy progression in type 2 diabetes. N Engl J Med 2010;363:233-44. [Crossref] [PubMed]
- Cooper-DeHoff RM, Gong Y, Handberg EM, et al. Tight blood pressure control and cardiovascular outcomes among hypertensive patients with diabetes and coronary artery disease. JAMA 2010;304:61-8. [Crossref] [PubMed]
- Elliott HL, Lloyd SM, Ford I, et al. Improving blood pressure control in patients with diabetes mellitus and high cardiovascular risk. Int J Hypertens 2010;2010.
- Ogihara T, Saruta T, Rakugi H, et al. Target blood pressure for treatment of isolated systolic hypertension in the elderly: Valsartan in elderly isolated systolic hypertension study. Hypertension 2010;56:196-202. [Crossref] [PubMed]
- Haller H, Ito S, Izzo JL Jr, et al. Olmesartan for the delay or prevention of microalbuminuria in type 2 diabetes. N Engl J Med 2011;364:907-17. [Crossref] [PubMed]
- Imai E, Chan JC, Ito S, et al. Effects of olmesartan on renal and cardiovascular outcomes in type 2 diabetes with overt nephropathy: A multicentre, randomised, placebo-controlled study. Diabetologia 2011;54:2978-86. [Crossref] [PubMed]
- Ruggenenti P, Fassi A, Ilieva AP, et al. Effects of verapamil added-on trandolapril therapy in hypertensive type 2 diabetes patients with microalbuminuria: The BENEDICT-B randomized trial. J Hypertens 2011;29:207-16. [Crossref] [PubMed]
- Ruggenenti P, Lauria G, Iliev IP, et al. Effects of manidipine and delapril in hypertensive patients with type 2 diabetes mellitus: The delapril and manidipine for nephroprotection in diabetes (DEMAND) randomized clinical trial. Hypertension 2011;58:776-83. [Crossref] [PubMed]
- Tillin T, Orchard T, Malm A, et al. The role of antihypertensive therapy in reducing vascular complications of type 2 diabetes. Findings from the DIabetic REtinopathy Candesartan Trials-Protect 2 study. J Hypertens 2011;29:1457-62. [Crossref] [PubMed]
- Zhang Y, Zhang X, Liu L, et al. Is a systolic blood pressure target <140 mmHg indicated in all hypertensives? Subgroup analyses of findings from the randomized FEVER trial. Eur Heart J 2011;32:1500-8. [Crossref] [PubMed]
- Parving HH, Brenner BM, McMurray JJ, et al. Cardiorenal end points in a trial of aliskiren for type 2 diabetes. N Engl J Med 2012;367:2204-13. [Crossref] [PubMed]
- Benavente OR, Coffey CS, Conwit R, et al. Blood-pressure targets in patients with recent lacunar stroke: The SPS3 randomised trial. Lancet 2013;382:507-15. [Crossref] [PubMed]
- Fried LF, Emanuele N, Zhang JH, et al. Combined angiotensin inhibition for the treatment of diabetic nephropathy. N Engl J Med 2013;369:1892-903. [Crossref] [PubMed]
- Win S, Anand I, McMurray J, et al. Morbidity and mortality in diabetics with heart failure and a preserved ejection fraction: Results from the i-preserve trial. J Am Coll Cardiol 2013;61:E706. [Crossref]
- Palacio S, McClure LA, Benavente OR, et al. Lacunar strokes in patients with diabetes mellitus: risk factors, infarct location, and prognosis: the secondary prevention of small subcortical strokes study. Stroke 2014;45:2689-94. [Crossref] [PubMed]
- Reboussin DM, Allen NB, Griswold ME, et al. Systematic Review for the 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol 2018;71:2176-98. [Crossref] [PubMed]
- Bangalore S, Kumar S, Lobach I, et al. Blood pressure targets in subjects with type 2 diabetes mellitus/impaired fasting glucose: observations from traditional and bayesian random-effects meta-analyses of randomized trials. Circulation 2011;123:2799-810. [Crossref] [PubMed]
- Aronow WS, Fleg JL, Pepine CJ, et al. ACCF/AHA 2011 expert consensus document on hypertension in the elderly: A report of the American college of cardiology foundation task force on clinical expert consensus documents. J Am Coll Cardiol 2011;57:2037-114. [Crossref] [PubMed]
- Mancia G, Fagard R, Narkiewicz K, et al. The task force for the management ofarterial hypertension of the european society ofhypertension (esh) and of the european society of cardiology (esc). J Hypertens 2013;31:1281-357. [Crossref] [PubMed]
- Aschner P, Beck-Nielsen H, Bennett P, et al. Global guideline for type 2 diabetes. Diabetes Res Clin Pract 2014;104:1-52. [Crossref] [PubMed]
- Go AS, Bauman MA, Coleman King SM, et al. An effective approach to high blood pressure control: A science advisory from the American Heart Association, the American College of Cardiology, and the Centers for Disease Control and Prevention. J Am Coll Cardiol 2014;63:1230-8. [Crossref] [PubMed]
- American Diabetes Association. 8. Cardiovascular disease and risk management. Diabetes Care 2015;38:S49-57. [Crossref] [PubMed]
- Introduction: Standards of Medical Care in Diabetes-2018. Diabetes Care 2018;41:S1-2. [Crossref] [PubMed]
- American Diabetes A. Standards of Medical Care in Diabetes-2018 Abridged for Primary Care Providers. Clin Diabetes 2018;36:14-37. [PubMed]
- American Diabetes A. 11. Older Adults: Standards of Medical Care in Diabetes-2018. Diabetes Care 2018;41:S119-25. [Crossref] [PubMed]