Physical activity levels are low in patients with pulmonary hypertension
Introduction
According to international recommendations, all adults should undertake ≥150 min/week of moderate physical activity (PA) or ≥75 min/week of vigorous-intensity PA, or an equivalent combination of the two [≥150 min/week of moderate-to-vigorous PA (MVPA)] for health promotion (1). To design effective PA interventions, PA levels need to be accurately assessed in the different population subsets (1). In this regard, limited data are available on objectively assessed PA levels in patients with pulmonary hypertension (PH), a disease commonly associated with severe physical capacity impairment (2-4).
We assessed PA levels via accelerometry in a cohort of Spanish PH patients. We established whether such levels complied with international PA recommendations and how they compared with those of PH-free controls. We also evaluated potential associations of meeting international PA recommendations with recently reported disease outcome predictors (5).
Methods
The study protocol received Institutional Review Board approval (approval number: 13/377) and adhered to the tenets of the Declaration of Helsinki. All participants provided written informed consent.
Controls (n=107) had been previously recruited by us (6). Inclusion criteria were as follows: aged 18–79 years, able to understand the requirements for valid accelerometry, and living in the Madrid southern area. The majority (70%) of this group had no major chronic disease (including cardiopulmonary disorders or cancer), whereas 30% had cardiometabolic diseases (dyslipidemia, obesity, arterial hypertension, diabetes). No subject had PH. Their PA levels were measured with the same accelerometers and methodology that is described below for patients.
Patients were recruited from the PH unit of the Hospital Universitario 12 de Octubre (also located in the southern Madrid suburbs) from March 2014–May 2016 if meeting the following criteria: outpatient hemodynamically diagnosed with PH and following treatment in the aforementioned unit, aged 18–75 years, able to walk independently and understand the requirements for valid accelerometry, and living in the southern Madrid area. Outcome assessment in this group was consistently performed in the same order (within the same day): 6-minute walk distance (6MWD) test (7), followed by distribution of triaxial accelerometers (8) to wear whilst awake (except for water activities) during 7–10 days.
A minimum of 5-day monitoring (including weekends), with a minimum of 10 h/day of complete accelerometry [GT3X monitor device (Actigraph; Pensacola, FL, USA)] was considered necessary per participant (6). This criterion was also used to evaluate adherence to accelerometer use by subjects. Data were analyzed using ActiLife5 LITE software (Actigraph). We determined average intensity (counts/min) by dividing the sum of the total counts per pre-defined epoch (15 s) for a valid day by the number of minutes of wear time in that day across all valid days. Thereafter, counts were converted to average daily time engaged in inactivity or different PA-intensities using previously defined cut-offs (9).
If previously assessed by us in the preceding 3 months, we also entered into the analyses patients’ data of blood N-terminal pro-B-type natriuretic peptide (NT-proBNP) or cardiopulmonary exercise testing [CET, i.e., peak oxygen uptake (VO2peak), and end-tidal pressure (PETCO2@AT), and ventilatory equivalent of carbon dioxide at the anaerobic threshold (VE/VCO2@AT)], as detailed elsewhere (10).
Accelerometry outliers outside the inter-quartile range were removed with box and whisker plots. We compared accelerometry data and % adherence to international PA guidelines between groups using unpaired Student’s t-test and Fisher’s exact test, respectively. In the PH group, comparisons between New York Heart Association (NYHA) or disease etiology categories were performed by one-factor ANOVA. Correlations between PA levels and age, body mass index (BMI), hemodynamic variables at rest, and CET variables were evaluated with Pearson’s correlation coefficient (r), where the strength of the correlation was assessed according to the following interpretation: “poor” (r=0.00–0.30), “moderate” (r=0.31–0.60), “moderately high” (r=0.61–0.84), and “high” (r≥0.85). Finally, logistic regression analyses were performed within this group stratified by likelihood of survival.
Results
Of 209 potentially eligible patients within the aforementioned period, and after exclusion of those not meeting the aforementioned inclusion criteria (n=75), declining participation (n=46) or not providing valid accelerometer data (n=12; adherence rate: 86% of the patients), and of 1 “outlier”, accelerometer data valid for analyses were available from 75 patients. Table 1 lists the main characteristics of the two groups.
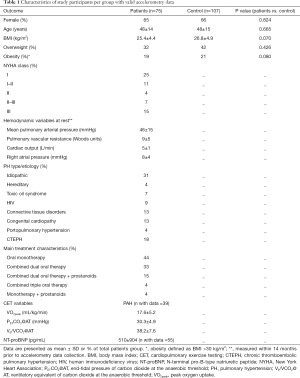
Full table
With the exception of vigorous PA (with very low values in both groups), all accelerometer data showed significant differences between patients and controls, with lower PA levels and proportion of individuals meeting PA guidelines, but higher inactivity time, in the former (Table 2). The results remained essentially unchanged when comparing patients by NHYA class or disease etiology versus controls, or PA variables within the patient group by NHYA class or etiology (data not shown). Only NYHA class III patients showed lower (P<0.001) total activity time (139±50 min/day) than their class I (271±101 min/day) or II–III referents (318±152 min/day).
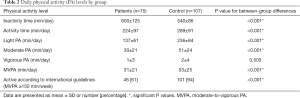
Full table
In the patients’ group, we found a significant, yet poor/moderate correlation between moderate PA and mean pulmonary arterial pressure (r=−0.254; P=0.037), as well as between inactivity time and BMI (r=−0.342; P=0.004). No other significant correlations were revealed. Additionally, MVPA levels or proportion of individuals meeting minimum international guidelines (MVPA ≥150 min/week) were significantly higher in those with higher likelihood of survival at 4 years according to previously reported (5) predictive threshold-values of VO2peak (≥15 mL/kg/min), 6MWD (≥464 m), PETCO2@AT (≥30 mmHg), VE/VCO2@AT (≤39) or NT-proBNP (≤914 pg/mL) (data not shown). The odds ratio (OR) of having a “low-risk” value of 6MWD (≥464 m) or VE/VCO2@AT (≤39) was ~4-fold higher in patients following MVPA guidelines than in their less active peers [see Figure 1; OR =4.3, 95% confidence interval (CI), 1.6–11.6, P=0.005 and OR =4.5, 95% CI, 0.9–21.1, P=0.054, respectively].
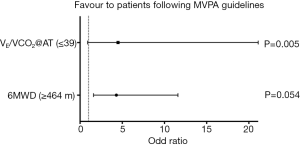
Discussion
In a representative, relatively large cohort of PH patients (accounting for ~15% of the total Spanish patient population), less than two-thirds of patients met the minimum recommended level of PA, and not meeting such guidelines was associated with a higher risk profile. Only few previous studies on smaller cohorts have used accelerometry to assess PA levels in PH patients. Matura et al. found MVPA levels well below 150 min/week in 15 women aged ~51 years on average (3). Also, Mainguy et al. found lower MVPA levels in patients with idiopathic PH (n=5) or with PH associated with limited systemic sclerosis (n=10) than in their controls (2). Using the same accelerometer as used in our study, Pugh et al. found also that inactivity time was significantly higher but levels of all PA-intensities were lower in 20 patients with PH than in 30 healthy controls (4). The main limitation of the present study could be the interpretation of the results, because they reflect an association and not a cause-effect relationship between PA level and disease progression. Further, the more affected patients are less likely to be motivated to be physically active. Another shortcoming is that accelerometers do not register “static” or indoor physical activities (e.g., indoor bicycling, swimming, weight lifting) and thus actual PA levels could be underestimated in these special populations.
Taken together, the present findings and those of previous research (2-4) suggest that daily PA is reduced in patients with PH to a level that may contribute to further compromise the exercise capacity of this population and even their odds of survival. Efforts should be made to promote the implementation of healthy PA habits in these patients and further research might determine how this intervention might impact disease outcomes, in line with previous interventional research (11,12).
Acknowledgements
A Lucia is funded by Fondo de Investigaciones Sanitarias (Instituto de Salud Carlos III) and Fondos Feder (grant #PI15/00558).
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The study protocol received Institutional Review Board approval (approval number: 13/377) and adhered to the tenets of the Declaration of Helsinki. All participants provided written informed consent.
References
- Global Recommendations on Physical Activity for Health. WHO Guidelines Approved by the Guidelines Review Committee. Geneva: World Health Organization, 2010.
- Mainguy V, Provencher S, Maltais F, et al. Assessment of daily life physical activities in pulmonary arterial hypertension. PLoS One 2011;6:e27993. [Crossref] [PubMed]
- Matura LA, Shou H, Fritz JS, et al. Physical Activity and Symptoms in Pulmonary Arterial Hypertension. Chest 2016;150:46-56. [Crossref] [PubMed]
- Pugh ME, Buchowski MS, Robbins IM, et al. Physical activity limitation as measured by accelerometry in pulmonary arterial hypertension. Chest 2012;142:1391-8. [Crossref] [PubMed]
- Quezada-Loaiza CA, Flox-Camacho A, Santos-Lozano A, et al. Predictive value of NT-proBNP combined with exercise capacity variables in pulmonary artery disease: Insights from a Spanish cohort. Int J Cardiol 2015;186:32-4. [Crossref] [PubMed]
- Ruiz-Casado A, Verdugo AS, Solano MJ, et al. Objectively assessed physical activity levels in Spanish cancer survivors. Oncol Nurs Forum 2014;41:E12-20. [Crossref] [PubMed]
- Guyatt GH, Pugsley SO, Sullivan MJ, et al. Effect of encouragement on walking test performance. Thorax 1984;39:818-22. [Crossref] [PubMed]
- Santos-Lozano A, Marín PJ, Torres-Luque G, et al. Technical variability of the GT3X accelerometer. Med Eng Phys 2012;34:787-90. [Crossref] [PubMed]
- Freedson PS, Melanson E, Sirard J. Calibration of the Computer Science and Applications, Inc. accelerometer. Med Sci Sports Exerc 1998;30:777-81. [Crossref] [PubMed]
- Sanchis-Gomar F, González-Saiz L, Sanz-Ayan P, et al. Rationale and Design of a Randomized Controlled Trial Evaluating Whole Muscle Exercise Training Effects in Outpatients with Pulmonary Arterial Hypertension (WHOLEi+12). Cardiovasc Drugs Ther 2015;29:543-50. [Crossref] [PubMed]
- Mereles D, Ehlken N, Kreuscher S, et al. Exercise and respiratory training improve exercise capacity and quality of life in patients with severe chronic pulmonary hypertension. Circulation 2006;114:1482-9. [Crossref] [PubMed]
- Weinstein AA, Chin LM, Keyser RE, et al. Effect of aerobic exercise training on fatigue and physical activity in patients with pulmonary arterial hypertension. Respir Med 2013;107:778-84. [Crossref] [PubMed]


