Effects of peep on lung injury, pulmonary function, systemic circulation and mortality in animals with uninjured lungs—a systematic review
Introduction
Artificial ventilation is a frequently applied intervention in critically ill patients in need of ventilatory support (1,2), and indispensable in patients under general anesthesia for surgery (3,4). One main side effect of artificial ventilation using positive pressure is alveolar collapse at the end of expiration (5,6). This could not only cause shunt and thus negatively affect arterial oxygenation, but also induce lung injury, through repetitive opening and closing of those lung parts that collapse at the end of expiration—a phenomenon frequently referred to as ventilator-induced lung injury (VILI) (7). Use of positive end-expiratory pressure (PEEP) could prevent alveolar collapse and therefore some level of PEEP is frequently chosen.
There is clear evidence for clinical benefit of ‘high PEEP’ in patients with acute respiratory distress syndrome (ARDS) (8). One metaanalysis that used individual patient data from three large randomized controlled trials (RCTs) of PEEP (9-11) showed ‘high PEEP’ to be associated with increased survival. However, it should also be acknowledged that mortality benefit of ‘high PEEP’ was restricted to patients with moderate or severe ARDS, and ‘high PEEP’ was even associated with prolonged duration of ventilation in patients classified as having mild ARDS. There is little evidence for benefit of PEEP, at any level, in ICU patients without ARDS (12), and in surgery patients receiving intraoperative ventilation (13). Despite these findings, observational studies show an increase in use of (higher levels of) PEEP in ICU patient without ARDS (1,2), as well as in surgery patients (4,14,15).
One important notion is that PEEP, at any level could cause overdistension of non-dependent lung parts (16-18). Alike repetitive opening and closing, overdistension is considered yet another causal factor for VILI (16,17). PEEP can also negatively impact systemic circulation through its effects on right ventricle loading conditions (19), and afterload (19,20). Indeed, one recent trial of PEEP in surgery patients showed much more episodes of shock during the surgical procedure with the use of higher levels of PEEP (13).
The balance between benefit and harm of (higher levels of) PEEP in the presence of lung injury thus may differ from that in the absence of lung injury. Nevertheless, we hypothesized that PEEP prevents against lung injury and improves pulmonary function, while not affecting non-pulmonary endpoints in mechanical ventilation models using animals with uninjured lungs We performed a systematic review of the literature to search for trials that compared different levels of PEEP in animals with uninjured lungs.
Methods
Search strategy
Two independent investigators performed an unrestricted search in the databases of PubMed and CENTRAL (the Cochrane Library) for relevant articles published up to January 2017, using the following Medical Subject Headings and keywords: ‘PEEP’ OR ‘positive end-expiratory pressure’ OR ‘positive-end expiratory pressure’ OR ‘positive end expiratory pressure’ AND ‘randomized’ OR ‘RCT’.
All articles returned by this search were screened for eligibility by reading the title and abstract. If considered potentially relevant, the full text was reviewed. References of all these articles as well as reviews and metaanalysis of PEEP were checked for potentially relevant articles missed by the search.
Selection of studies
The single one inclusion criterion was that animals had to be randomized to different levels of PEEP.
The following exclusion criteria were used: (I) animals were subjected to a ‘hit’ causing lung injury, either before or during ventilation, consisting of (repeated) lung lavage(s), administration of oleic acid, or infection; (II) ‘bundles’ of ventilation were compared, meaning that animals were randomized to ventilation with different PEEP levels but also differences in other ventilator settings, like tidal volume (VT) size; (III) when more than one PEEP level was used within an individual animal; and finally (IV) when the trial used immature animals. In case of more than two randomization groups within one trial, we focused one the two groups with the highest and the lowest level of PEEP.
Data extraction
The two investigators extracted the data into a database constructed for this investigation. Any disagreement between investigators on the data extracted was solved by discussion. The following data were extracted: type of animal, age of animal, gender of animal, duration of ventilation before randomization, duration of ventilation, ventilator settings including ventilation mode, tidal volume size, level of PEEP, peak and plateau pressures, fraction of inspired oxygen (FiO2), pulmonary compliance and if present blood gas analysis results, hemodynamic parameters as reported, such as blood pressure, heart rate, cardiac output or index, and final outcome (e.g., death, if not by sacrifice).
Quality assessment
The same two investigators independently performed quality assessment of the included trials. The SYRCLE’s Risk of Bias tool was used to assess the risk of bias of all included studies (21), where again disagreements were settled by discussion.
Definitions
As the set levels of PEEP were quite different between trials, we decided to use the following wording for PEEP levels: ‘high PEEP’ was used for the PEEP level for trials that compared a higher level of PEEP to a low level of PEEP, where low PEEP was referred to as ‘low PEEP’; ‘PEEP’ was used for trials that compared some level of PEEP to no PEEP, where no PEEP was referred to as ‘no PEEP’.
Outcomes
The primary outcome of interest was occurrence or severity of lung injury at the end of the experiment, as assessed and reported by the investigators of the individual trial, which could be histopathology by conventional microscopy or electron microscopy, lung weight or wet-to-dry ratios, or visual inspection. Secondary outcomes included: (I) variables reflecting pulmonary function (e.g., arterial oxygenation, the respiratory system compliance); (II) hemodynamic outcomes (e.g., heart rate, arterial blood pressure, cardiac output or index, and plasma lactate levels) and; (III) mortality at the end of the experiment, if not by sacrifice.
Results
Search results
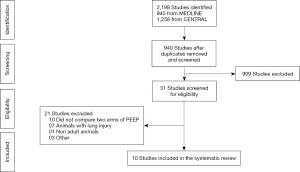
The search returned 2,198 articles, 940 from MEDLINE and 1,258 from CENTRAL (Figure 1). After removal of duplicates, 940 articles were evaluated by reading title and abstracts. Most articles, 909 in total, did not meet the inclusion criterion. After carefully assessment of full text of the remaining 31 potentially eligible articles, 21 were excluded for the following reasons: (I) did not compare different PEEP levels (n=10); (II) ventilation in animals with existing lung injury (n=7); (III) immature animals (n=1); and (IV) other reasons (n=3). Thus, 10 trials including 284 animals remained (22-31). Table 1 summarizes characteristics of the included trials. Three trials used rats (23,25,28), three used pigs (26,27,30), two used rabbits (24,29), one used dogs (22) and one used horses (31). The median included animals were 8 in both PEEP arms.

Full table
Quality trials
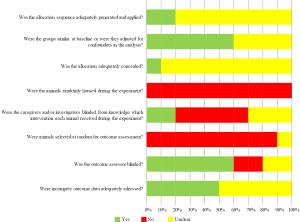
For the majority of the trials the risk of bias is unclear, since details of the trials were insufficiently provided to assess the risk of bias properly (Figure 2). The high risk of bias was in the domains related to housing of the animals, randomly selection of the animals for outcome assessment and blinding of the caregivers and/or investigator to which intervention each animal received during the experiment.
Ventilation characteristics
Before randomization, in three trials a volume-controlled mode was used (22,26,27), in two a pressure-controlled mode (23,30), and in one a spontaneous breathing mode (31). After randomization, in four trials a volume-controlled mode was used (22,26,27,31), and in two a pressure-controlled mode (23,30). For four trials it remained unclear which ventilation mode was used (24,25,28,29). Duration of ventilation varied widely, from 1 to 6 hours (Table 1). In four trials, recruitment maneuvers were used in the ‘high PEEP’ or ‘PEEP’ arms (22-25), in three trials recruitment maneuvers were used in both randomization arms (22,23,25). There was a noticeable variation in tidal volume size, from 7 to as high as 60 mL/kg, though tidal volume size was always similar in the ‘high PEEP’ or ‘PEEP’ arms and ‘low PEEP’ or ‘no PEEP’ arms in the individual trials. Tidal volume size was not reported in three trials (23,26,27).
The levels of PEEP compared
PEEP was most often 10 cmH2O in the ‘high PEEP’ or ‘PEEP’ arms (22,23,26,30), and most often 0 cmH2O in the ‘low PEEP’ or ‘no PEEP’ arms (Table 1) (23,25-28). Interestingly, in three trials PEEP was 5 cmH2O in the ‘high PEEP’ or ‘PEEP’ arms (25,27,28), while in three other trials the same level of PEEP was used in the ‘low PEEP’ arms (29-31). In all trials, PEEP was fixed, except in one trial in which PEEP was titrated from 5 up to 20 cmH2O in 15 minutes intervals in the ‘high PEEP’ arm (31).
Outcomes
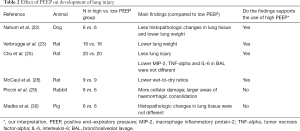
Lung injury (Table 2)—of the six trials investigating the effects of PEEP on lung injury, four showed benefit (22,23,25,28) and two trials showed no benefit in animals ventilated with ‘high PEEP’ or ‘PEEP’ (29,30).
Full table
All three trials comparing ‘PEEP’ with ‘no PEEP’ showed less VILI with ‘PEEP’ (23,25,28). Of the three trials comparing ‘high PEEP’ to ‘low PEEP’, one trial showed less VILI with ‘high PEEP’ (22), while one found more VILI (29) and another found no differences between the PEEP arms (30).
Pulmonary function (Table 3)—of the nine trials investigating the effects of PEEP on the lung parameters, seven trials showed better arterial oxygenation or improved respiratory system compliance with ‘high PEEP’ or ‘PEEP’ (23,24,26-28,30,31), while two showed no benefit (22,29).

Full table
Of the four trials comparing ‘PEEP’ with ‘no PEEP’ (23,26-28), all showed better arterial oxygenation or improved respiratory system compliance in the ‘PEEP’ arms. Five trials compared ‘high PEEP’ with ‘low PEEP’, of which three trials showed improved pulmonary outcomes in the ‘high PEEP’ arms (24,30,31), and one showed a worsened pulmonary function (29) and another found no differences (22).
Hemodynamic outcomes (Table 4)—of the seven trials that reported on hemodynamic outcomes, in six trials ‘high PEEP’ or ‘PEEP’ resulted in a lower cardiac output, hypotension or hyperlactatemia (26-31), while one trial showed no harm (22).

Full table
All of the three trials comparing ‘PEEP’ with ‘no PEEP’ showed lower cardiac output, hypotension or hyperlactatemia with ‘PEEP’ (26-28). Of the four trials comparing ‘high PEEP’ with ‘low PEEP’, three showed negative effects of ‘high PEEP’ on the hemodynamic variables (29-31), while one showed no harm (22).
Mortality (Table 5)—of the five trials that reported on mortality, two showed higher mortality rates with ‘high PEEP’ or ‘PEEP’ (26,27), two trials showed lower mortality rates with ‘PEEP’ or ‘high PEEP’ (22,28), and one trial found no differences (30).

Full table
Of the three trials comparing ‘PEEP’ to ‘no PEEP’ two showed higher mortality rates (26,27), while the other one showed a lower mortality rate with ‘PEEP’ (28). Of the two trials comparing ‘high PEEP’ with ‘low PEEP’, one showed lower mortality rates (22), while the other found no differences (30).
Discussion
The results of this systematic review of trials comparing ‘high PEEP’ or ‘PEEP’ with ‘low PEEP’ or ‘no PEEP’ in animals with uninjured lungs can be summarized as follows: (I) the number of trials evaluating the effects of PEEP is severely limited; (II) trials are heterogeneous in design and outcomes; (III) the beneficial effect of PEEP on VILI was not found in all trials; (IV) PEEP, at any level, has a clear beneficial effect on pulmonary physiology; (V) PEEP negatively effects systemic circulation; and (VI) the effect of PEEP on mortality is diverse.
To our best knowledge this is the first systematic review investigating the effects of ventilation with different levels of PEEP in animals with uninjured lungs. Strength of this systematic review is that we restricted inclusion to trials that only compared two different levels of PEEP, i.e., trials that compared so-called ‘bundles of protective ventilation’ were excluded. As such we minimized the risk of confounding effects e.g., differences in tidal volume size. Also, the search strategy was very wide and found a small but reasonable number of trials. Finally, the included trials assessed different types of animals, which could increase generalizability of the findings.
One possible explanation for the fact that some trials showed less lung injury with ‘high PEEP’ or ‘PEEP’ while others showed no benefit with respect to development of VILI could be the wide variation of definitions for VILI used. It should also be noticed that some trials used extremely large tidal volumes, high peak inspiratory pressures and plateau pressures, or high FiO2, which are nowadays considered unsafe. Also, in some trials animals were exposed to long cold ischemic time, or high dosages of epinephrine. It remains uncertain if PEEP has the potential to protect against lung injury under such extreme conditions. Actually, one might wonder if these animals were not having lung injury, but if lung injury was induced during the trial.
One clear finding was that in almost all trials ‘high PEEP’ or ‘PEEP’ resulted in better physiologic endpoints, like arterial oxygenation and respiratory system compliance. These findings are very consistent with observations in clinical trials of PEEP, both in trials in ICU patients with ARDS (9-11), and ICU patients without ARDS (32,33). Another clear finding, though, was that ventilation with PEEP negatively affects systemic circulation. Positive pressure ventilation, per se, increases the intrathoracic pressure, decreasing preload and increasing afterload of the right ventricle (34), while it can reduce the afterload of the left ventricle. The present findings echo the results of one recent metaanalysis of clinical trials that compared different levels of PEEP (35), and also the clear finding that PEEP induces shock in one recent clinical trial of intraoperative ventilation (13).
The results of this systematic review add to our knowledge on the effects of PEEP during ventilation in the absence of lung injury. Its results are also in line with those from a recent systematic review and metaanalysis of clinical trials investigating the effects of PEEP in patients without ARDS (35). That investigation suggested no benefit of ventilation with (higher levels of) PEEP with regard to important clinical endpoints, like mortality and duration of mechanical ventilation. That investigation, though, also suggested improved pulmonary function with (higher levels of) PEEP, like a better oxygenation, and there was at least a suggestion that it could negatively affect systemic circulation. These two systematic reviews underline the need for well-conducted and sufficiently sized clinical, and animal trials investigating the effects of PEEP in patients without ARDS, and in animals without uninjured lungs.
This systematic review has several limitations. First, it should be noted that the trials identified by the search maybe better reflect the clinical scenario of intraoperative ventilation, seen the relative short period of ventilation in all of them. On the other hand, one could also argue that duration of ventilation in the trials reviewed here is very similar to trials of PEEP in animals with lung injury, and these trials have been frequently used in the translation of effects of PEEP from animals to patients with ARDS. Second, the overall quality of the included trials, as with all systemic reviews, influenced the results. The majority of the included trials had an unclear or high risk of bias, and in addition sample sizes were small and not all outcomes were reported in all trials. Third, similar levels of PEEP served as ‘high PEEP’ or ‘low PEEP’ across the trials, further complicating the interpretation of the findings. Finally, and as mentioned above, in some trials ventilator settings other than PEEP are by now no longer accepted as ‘safe’.
Conclusions
The number of trials comparing different levels of PEEP in animals with uninjured lungs is limited and the results of these trials are difficult to compare. Based on the findings of this systematic review it remains uncertain whether PEEP, at any level, prevents lung injury in animals with uninjured lungs. While (higher levels of) PEEP improves pulmonary function, it also negatively affects systemic circulation.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Esteban A, Frutos-Vivar F, Muriel A, et al. Evolution of mortality over time in patients receiving mechanical ventilation. Am J Respir Crit Care Med 2013;188:220-30. [Crossref] [PubMed]
- Esteban A, Ferguson ND, Meade MO, et al. Evolution of mechanical ventilation in response to clinical research. Am J Respir Crit Care Med 2008;177:170-7. [Crossref] [PubMed]
- Futier E, Godet T, Millot A, et al. Mechanical ventilation in abdominal surgery. Ann Fr Anesth Reanim 2014;33:472-5. [Crossref] [PubMed]
- Güldner A, Kiss T, Serpa Neto A, et al. Intraoperative Protective mechanical ventilation for prevention of postoperative pulmonary complications. Anesthesiology 2015;123:692-713. [Crossref] [PubMed]
- Dries DJ. Assisted ventilation. J Burn Care Res 2016;37:75-85. [Crossref] [PubMed]
- Bendixen HH, Hedley-Whyte J, Laver MB. Impaired oxygenation in surgical patients during general anesthesia with controlled ventilation. A concept of atelectasis. N Engl J Med 1963;269:991-6. [Crossref] [PubMed]
- Slutsky AS, Ranieri VM. Ventilator-induced lung injury. N Engl J Med 2014;370:980. [PubMed]
- Briel M, Meade M, Mercat A, et al. Higher vs lower positive end-expiratory pressure in patients with acute lung injury and acute respiratory distress syndrome: systematic review and meta-analysis. JAMA 2010;303:865-73. [Crossref] [PubMed]
- Brower RG, Lanken PN, MacIntyre N, et al. Higher versus lower positive end-expiratory pressures in patients with the acute respiratory distress syndrome. N Engl J Med 2004;351:327-36. [Crossref] [PubMed]
- Meade MO, Cook DJ, Guyatt GH, et al. Ventilation strategy using low tidal volumes, recruitment maneuvers, and high positive end-expiratory pressure for acute lung injury and acute respiratory distress syndrome: a randomized controlled trial. JAMA 2008;299:637-45. [Crossref] [PubMed]
- Mercat A, Richard JCM, Vielle B, et al. Positive end-expiratory pressure setting in adults with acute lung injury and acute respiratory distress syndrome: a randomized controlled trial. JAMA 2008;299:646-55. [Crossref] [PubMed]
- Serpa Neto A, Simonis FD, Schultz MJ. How to ventilate patients without acute respiratory distress syndrome? Curr Opin Crit Care 2015;21:65-73. [Crossref] [PubMed]
- PROVE Network Investigators for the Clinical Trial Network of the European Society of Anaesthesiology, Hemmes SNT, Gama de Abreu M, et al. High versus low positive end-expiratory pressure during general anaesthesia for open abdominal surgery (PROVHILO trial): a multicentre randomised controlled trial. Lancet 2014;384:495-503. [Crossref] [PubMed]
- Fernández-Pérez ER, Sprung J, Afessa B, et al. Intraoperative ventilator settings and acute lung injury after elective surgery: a nested case control study. Thorax 2009;64:121-7. [Crossref] [PubMed]
- Hess DR, Kondili D, Burns E, et al. A 5-year observational study of lung-protective ventilation in the operating room: a single-center experience. J Crit Care 2013;28:533.e9-15. [Crossref] [PubMed]
- Bellardine Black CL, Hoffman AM, Tsai LW, et al. Relationship between dynamic respiratory mechanics and disease heterogeneity in sheep lavage injury. Crit Care Med 2007;35:870-8. [Crossref] [PubMed]
- Samary CS, Santos RS, Santos CL, et al. Biological impact of transpulmonary driving pressure in experimental acute respiratory distress syndrome. Anesthesiology 2015;123:423-33. [Crossref] [PubMed]
- Gama de Abreu M, Cuevas M, Spieth PM, et al. Regional lung aeration and ventilation during pressure support and biphasic positive airway pressure ventilation in experimental lung injury. Crit Care 2010;14:R34. [Crossref] [PubMed]
- Luecke T, Pelosi P. Clinical review: Positive end-expiratory pressure and cardiac output. Crit Care 2005;9:607-21. [Crossref] [PubMed]
- Putensen C, Mutz NJ, Putensen-Himmer G, et al. Spontaneous breathing during ventilatory support improves ventilation-perfusion distributions in patients with acute respiratory distress syndrome. Am J Respir Crit Care Med 1999;159:1241-8. [Crossref] [PubMed]
- Hooijmans CR, Rovers MM, de Vries RB, et al. SYRCLE's risk of bias tool for animal studies. BMC Med Res Methodol 2014;14:43. [Crossref] [PubMed]
- Nahum A, Hoyt J, Schmitz L, et al. Effect of mechanical ventilation strategy on dissemination of intratracheally instilled Escherichia coli in dogs. Crit Care Med 1997;25:1733-43. [Crossref] [PubMed]
- Verbrugge SJ, Šorm V, van 't Veen A, et al. Lung overinflation without positive end-expiratory pressure promotes bacteremia after experimental Klebsiella pneumoniae inoculation. Intensive Care Med 1998;24:172-7. [Crossref] [PubMed]
- Mols G, Hermle G, Fries G, et al. Different strategies to keep the lung open: a study in isolated perfused rabbit lungs. Crit Care Med 2002;30:1598-604. [Crossref] [PubMed]
- Chu EK, Whitehead T, Slutsky AS. Effects of cyclic opening and closing at low- and high-volume ventilation on bronchoalveolar lavage cytokines. Crit Care Med 2004;32:168-74. [Crossref] [PubMed]
- Krismer AC, Wenzel V, Lindner KH, et al. Influence of positive end-expiratory pressure ventilation on survival during severe hemorrhagic shock. Ann Emerg Med 2005;46:337-42. [Crossref] [PubMed]
- Herff H, Paal P, von Goedecke A, et al. Influence of ventilation strategies on survival in severe controlled hemorrhagic shock. Crit Care Med 2008;36:2613-20. [Crossref] [PubMed]
- McCaul C, Kornecki A, Engelberts D, et al. Positive end-expiratory pressure improves survival in a rodent model of cardiopulmonary resuscitation using high-dose epinephrine. Anesth Analg 2009;109:1202-8. [Crossref] [PubMed]
- Piccin VS, Calciolari C, Yoshizaki K, et al. Effects of different mechanical ventilation strategies on the mucociliary system. Intensive Care Med 2011;37:132-40. [Crossref] [PubMed]
- Madke GR, Forgiarini LA, Grün G, et al. Effect of positive end-expiratory pressure after porcine unilateral left lung transplant. Exp Clin Transplant 2013;11:50-5. [Crossref] [PubMed]
- Ambrósio AM, Ida KK, Souto MT, et al. Effects of positive end-expiratory pressure titration on gas exchange, respiratory mechanics and hemodynamics in anesthetized horses. Vet Anaesth Analg 2013;40:564-72. [Crossref] [PubMed]
- Michalopoulos A, Anthi A, Rellos K, et al. Effects of positive end-expiratory pressure (PEEP) in cardiac surgery patients. Respir Med 1998;92:858-62. [Crossref] [PubMed]
- Manzano F, Fernández-Mondéjar E, Colmenero M, et al. Positive-end expiratory pressure reduces incidence of ventilator-associated pneumonia in nonhypoxemic patients. Crit Care Med 2008;36:2225-31. [Crossref] [PubMed]
- Jellinek H, Krafft P, Fitzgerald RD, et al. Right atrial pressure predicts hemodynamic response to apneic positive airway pressure. Crit Care Med 2000;28:672-8. [Crossref] [PubMed]
- Serpa Neto A, Filho RR, Cherpanath T, et al. Associations between positive end-expiratory pressure and outcome of patients without ARDS at onset of ventilation: a systematic review and meta-analysis of randomized controlled trials. Ann Intensive Care 2016;6:109. [Crossref] [PubMed]